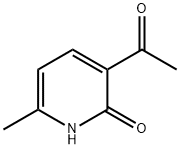
3-acetyl-6-methylpyridin-2(1H)-one synthesis
- Product Name:3-acetyl-6-methylpyridin-2(1H)-one
- CAS Number:25957-23-7
- Molecular formula:C8H9NO2
- Molecular Weight:151.16

75-16-1
264 suppliers
$12.00/10ml
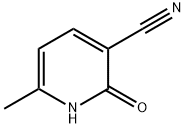
4241-27-4
220 suppliers
$6.00/250mg

25957-23-7
13 suppliers
inquiry
Yield:25957-23-7 53 %
Reaction Conditions:
in tetrahydrofuran at -78 - 20;
Steps:
34.A Step A: Synthesis of 3-acetyl-6-methylpyridin-2(1H)-one
At minus 78 degrees Celsius, add methylmagnesium bromide dropwise to tetrahydrofuran (20 ml) containing 6-methyl-2-oxo-1,2-dihydropyridine-3-carbonitrile (500 mg, 3.73 mmol) in tetrahydrofuran (5.47 ml, 1 mol per ml) was slowly raised to room temperature for 3 hours.After the reaction was completed, quenched with water, extracted with a mixed solution of dichloromethane and isopropanol (dichloromethane/isopropanol=3/1, 30 ml × 5 times), combined the organic phases, saturated brine (30 ml × 2 times) ), dried over anhydrous sodium sulfate, and concentrated.The resulting residue was purified by silica gel column chromatography (eluent: dichloromethane/methanol=10/1) to obtain 300 mg of pale yellow solid 3-acetyl-6-methylpyridin-2(1H)-one (yield rate: 53%).
References:
WO2023/16535,2023,A1 Location in patent:Page/Page column 45

5977-14-0
170 suppliers
$48.00/100g
51731-17-0
61 suppliers
$84.00/5g

25957-23-7
13 suppliers
inquiry
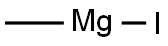
917-64-6
146 suppliers
$45.00/10ml

4241-27-4
220 suppliers
$6.00/250mg

25957-23-7
13 suppliers
inquiry