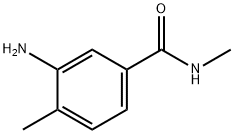
3-AMINO-N,4-DIMETHYLBENZAMIDE synthesis
- Product Name:3-AMINO-N,4-DIMETHYLBENZAMIDE
- CAS Number:54884-19-4
- Molecular formula:C9H12N2O
- Molecular Weight:164.2
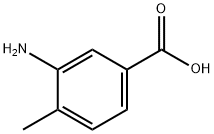
2458-12-0
320 suppliers
$15.00/5g

2592-95-2
649 suppliers
$14.14/1gm:

54884-19-4
24 suppliers
$49.00/200μmol
Yield:54884-19-4 95%
Reaction Conditions:
with 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride;N-ethyl-N,N-diisopropylamine;methylamine in tetrahydrofuran;DMF (N,N-dimethyl-formamide) at 54; for 2 h;
Steps:
2.1 Example 2 Step A
Example 2 Step A: A solution of 3-AMINO-4-METHYLBENZOIC acid (5.0 g, 33.1 mmol), EDC (12.7 g, 66.2 MMOL), N-hydroxybenzotriazole (5.37g, 39.72 mmol), a 2 M solution of methyl amine in THF (66 mL, 132.3 mmol), and diisopropylethyl amine (22.7 ML, 132.4 mmol) in THF (85 ML) and DMF (15 mL) was stirred at 54°C for 2 h. The mixture was concentrated under reduced pressure in vacuo, and the residue was purified by chromatography on a silica gel column. Elution with 20% EtOAc in hexanes, followed by EtOAc, and 2% MeOH in EtOAc afforded compound 2A as a cream- colored solid (5.14 g, 95 %). HPLC Retention time = 0.356 min.; LC/MS (M+H) + = 165.2.
References:
WO2003/99820,2003,A1 Location in patent:Page 35; 36
329940-36-5
8 suppliers
inquiry

54884-19-4
24 suppliers
$49.00/200μmol

96-98-0
347 suppliers
$8.00/10g

54884-19-4
24 suppliers
$49.00/200μmol
10397-30-5
88 suppliers
$58.20/10ml

54884-19-4
24 suppliers
$49.00/200μmol