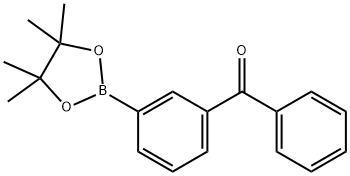
3-Benzoylphenylboronic acid pinacol ester synthesis
- Product Name:3-Benzoylphenylboronic acid pinacol ester
- CAS Number:949022-45-1
- Molecular formula:C19H21BO3
- Molecular Weight:308.18
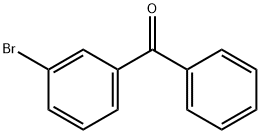
1016-77-9
180 suppliers
$10.00/1g
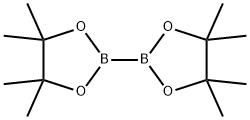
73183-34-3
573 suppliers
$6.00/5g

949022-45-1
33 suppliers
$67.00/250mg
Yield:949022-45-1 93%
Reaction Conditions:
with dichloro(1,1'-bis(diphenylphosphanyl)ferrocene)palladium(II)*CH2Cl2;potassium acetate in 1,4-dioxane at 80; for 12 h;Inert atmosphere;
Steps:
88.1 Step 1: phenyl(3-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl)methanone (478-2)
A mixture of Compound 478-1 (1.0 g, 3.8 mmol), BPD (1.5 g, 5.9 mmol) Pd(dppf)Cl2.CH2Cl2,(313 mg, 383 μmol), KOAc (1.1 g, 11.5 mmol) in dioxane (30 mL) was degassed and purged with N2 for 3 times, and then the mixture was stirred at 80 °C for 12 hrs under N2atmosphere. LCMS showed the reaction was completed. The reaction mixture was poured into H2O (30 mL), extracted with EtOAc (20 mL x 5). The combined organic layers were dried over Na2SO4, filtered and concentrated under reduced pressure to give a residue, which was purified by column chromatography (Petroleum ether / Ethyl acetate) to give Compound 478-2 (1.1 g, 3.6 mmol, 93% yield) as a light yellow solid.1H NMR (400MHz, CHLOROFORM-d) d = 8.23 (s, 1H), 8.03 (d, J=7.3 Hz, 1H), 7.87 (d, J=7.8 Hz, 1H), 7.81 (d, J=7.2 Hz, 2H), 7.64 - 7.56 (m, 1H), 7.53 - 7.45 (m, 3H), 1.36 (s, 12H).
References:
WO2021/3295,2021,A1 Location in patent:Paragraph 0730

1016-78-0
142 suppliers
$7.00/5g
25015-63-8
222 suppliers
$22.39/5G

119-61-9
782 suppliers
$5.00/10g

949022-45-1
33 suppliers
$67.00/250mg

579-18-0
75 suppliers
$40.00/2g

73183-34-3
573 suppliers
$6.00/5g

949022-45-1
33 suppliers
$67.00/250mg