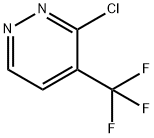
3-chloro-4-(trifluoromethyl)pyridazine synthesis
- Product Name:3-chloro-4-(trifluoromethyl)pyridazine
- CAS Number:749258-96-6
- Molecular formula:C5H2ClF3N2
- Molecular Weight:182.53
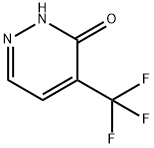
749258-95-5
27 suppliers
inquiry

749258-96-6
41 suppliers
inquiry
Yield:749258-96-6 61%
Reaction Conditions:
with N,N-dimethyl-formamide;trichlorophosphate at 120; for 2 h;
Steps:
Intermediate 49b: 3-chloro-4-(trifluoromethyl)pyridazine
[00514] Intermediate 49b: 3-chloro-4-(trifluoromethyl)pyridazine[00515] To a solution of 5-(trifluoromethyl)-1H-pyridazin-6-one (0.76g, 4.64mmol) in phosphorus oxychloride (4.33mL, 46.44mmol) was added a drop of N,N-dimethylformamide. The mixture washeated to 120 °C for 2 hours. The reaction mixture was allowed to cool to room temperature and concentrated in vacuo. The residue was diluted with EtOAc (lOOmL) and washed with water (lOOmL) and brine (lOOmL). The organic layer was dried over Na2504 and concentrated in vacuoto give 3-chloro-4-(trifluoromethyl)pyridazine (0.51g, 2.82mmol, 61% yield) as a brown oil which was used in the next step without further purification.MS Method 2: RT: 1.29 mi mlz 182.9 [M+H]
References:
WO2016/51193,2016,A1 Location in patent:Paragraph 00511; 00514; 00515
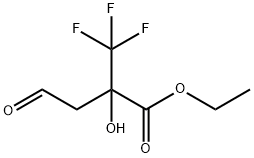
444689-44-5
0 suppliers
inquiry

749258-96-6
41 suppliers
inquiry