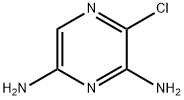
3-Chloro-pyrazine-2,6-diaMine synthesis
- Product Name:3-Chloro-pyrazine-2,6-diaMine
- CAS Number:794497-98-6
- Molecular formula:C4H5ClN4
- Molecular Weight:144.56
1458-01-1
201 suppliers
$5.00/1g

794497-98-6
16 suppliers
$110.00/50mg
Yield:794497-98-6 82%
Reaction Conditions:
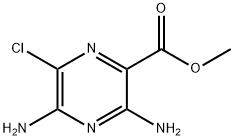
Stage #1: 3,5-diamino-6-chloro-pyrazine-2-carboxylic acid methyl esterwith lithium hydroxide;water in methanol at 90; for 1.5 h;
Stage #2: with hydrogenchloride in 1,4-dioxane;water at 100; for 2 h;
Stage #3: with sodium carbonate in water; pH=8;
Steps:
14
Preparation 14 3-Chloro-pyrazine-2,6-diamineLithium hydroxide (12.4g, 0.30 mol) was added to a stirred suspension of 3,5-diamino- 6-chloro-pyrazine-2-carboxylic acid methyl ester (20 g, 99 mmol) in methanol (300 ml) and water (120 ml) and the reaction heated at 9O0C for 1.5 hours before allowing to cool to room temperature. The reaction was concentrated in vacuo to afford a yellow slurry and this was suspended in 1 ,4-dioxane (350 ml) and 2M aqueous HCI solution (200 ml,) was added. The mixture was heated at 100 0C for 2 hours and then allowed to cool before removing the 1 ,4-dioxane in vacuo. The resulting aqueous solution was taken to pH 8 using sodium carbonate (saturated aqueous) and extracted into ethyl acetate (3 x 300 ml). The combined organic layers were washed with brine (300 ml), dried (Na2SO4) and concentrated in vacuo to afford a yellow solid (11.7g, 82%).1HNMR(d6-DMSO): 5.95(br s, 2H), 6.02(br s, 2H), 6.82(s, 1 H).
References:
WO2008/135830,2008,A1 Location in patent:Page/Page column 61