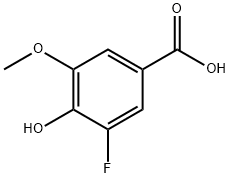
3-fluoro-4-hydroxy-5-methoxybenzoic acid synthesis
- Product Name:3-fluoro-4-hydroxy-5-methoxybenzoic acid
- CAS Number:773870-27-2
- Molecular formula:C8H7FO4
- Molecular Weight:186.14

79418-78-3
127 suppliers
$45.00/250mg

773870-27-2
9 suppliers
inquiry
Yield:773870-27-2 100%
Reaction Conditions:
with sodium chlorite;SULFAMIDE in 1,4-dioxane;water; for 0.5 h;Cooling with ice;
Steps:
80.2 Step (2): compound ii-15b → compound ii-15d
Amide sulfuric acid (48.7 g, 501 mmol) and water (750 mL) were added to asolutionofcompoundii-15b (38.8g, 228mmol) indioxane (750 mL). Theresultant solution was stirred while cooled with ice. To the solution was added sodium chlorite (45.3 g, 501 mmol). While cooled with ice, the solution was stirred for 30 minutes, and then thereto was dropwise added a 25% aqueous sodium hydrogensulfite solution (400 mL). Next, the resultant was subjected to extraction with ethyl acetate, and the organic phase was washed with a saturated saltsolution. The solution was dried over magnesium sulfate. Magnesium sulfate was filtrated off, and then the solution was concentrated. The residue was dried under reduced pressure to yield compound ii-15d (42.6 g, yield: 100%). 1H-NMR (DMSO-d6) δ: 10.08 (1H, s), 7.33-7.29 (2H, m), 3.85 (3H, s).
References:
EP2557082,2013,A1 Location in patent:Paragraph 0343; 0346
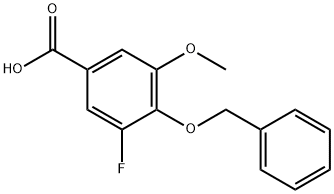
1078726-18-7
2 suppliers
inquiry

773870-27-2
9 suppliers
inquiry
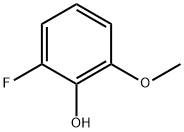
73943-41-6
98 suppliers
$13.00/250mg

773870-27-2
9 suppliers
inquiry