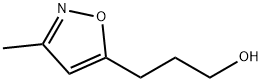
3-METHYL-5-ISOXAZOLEPROPANOL synthesis
- Product Name:3-METHYL-5-ISOXAZOLEPROPANOL
- CAS Number:105658-49-9
- Molecular formula:C7H11NO2
- Molecular Weight:141.17
75-21-8
215 suppliers
$39.10/1mL

300-87-8
242 suppliers
$6.00/5g

105658-49-9
50 suppliers
$108.00/1g
Yield:105658-49-9 86%
Reaction Conditions:
Stage #1: 3,5-dimethyl-1,2-oxazolewith n-butyllithium in tetrahydrofuran;hexane at -78; for 2 h;Inert atmosphere;
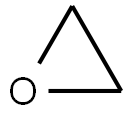
Stage #2: oxirane in tetrahydrofuran;hexane; for 1 h;Concentration;Temperature;
Steps:
74.1 3-(3-Methylisoxazol-5-yl)propan-1-ol
3,5-Dimethylisoxazole (4.00 g, 41.2 mmol) was dissolved in anhydrous tetrahydrofuran (50 mL). Under nitrogen atmosphere, n-butyllithium (16.5 mL, 2.5 M in n-heptane, 41.2 mmol) was slowly added at -78°C. The reaction wasstirred at -78°C for 2 hours, and added with ethylene oxide (1.81 g, 41.2 mmol) to continue to react for 1 hour. Thereaction was quenched by addition of saturated ammonium chloride solution (50 mL) and extracted with ethyl acetate(20 mL x 2). The organic phases were combined, washed with saturated sodium chloride solution (50 mL x 2), driedover anhydrous magnesium sulfate, filtered and the filtrate was concentrated under reduced pressure to give 3-(3-methylisoxazol-5-yl)propan-1-ol (5.00 g, as a yellow oil) with a yield of 86%. 1H NMR: (400 MHz, Methanol-d4) δ 6.05(s,1H), 3.63-3.55(m, 2H), 2.85-2.81(m, 2H), 2.27(s, 3H), 1.95-1.87(m, 2H). MS-ESI calcd. [M + H]+ 142, found 142
References:
EP3299371,2018,A1 Location in patent:Paragraph 0175; 0176; 0493; 0494

300-87-8
242 suppliers
$6.00/5g

105658-49-9
50 suppliers
$108.00/1g