
2-(2-CHLORO-ACETYLAMINO)-4,5,6,7-TETRAHYDRO-BENZO[B]THIOPHENE-3-CARBOXYLIC ACID METHYL ESTER synthesis
- Product Name:2-(2-CHLORO-ACETYLAMINO)-4,5,6,7-TETRAHYDRO-BENZO[B]THIOPHENE-3-CARBOXYLIC ACID METHYL ESTER
- CAS Number:300559-61-9
- Molecular formula:C12H14ClNO3S
- Molecular Weight:287.76
![2-AMINO-4,5,6,7-TETRAHYDRO-BENZO[B]THIOPHENE-3-CARBOXYLIC ACID METHYL ESTER](/CAS/GIF/108354-78-5.gif)
108354-78-5
86 suppliers
$6.00/250mg
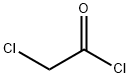
79-04-9
354 suppliers
$12.00/5g
![2-(2-CHLORO-ACETYLAMINO)-4,5,6,7-TETRAHYDRO-BENZO[B]THIOPHENE-3-CARBOXYLIC ACID METHYL ESTER](/StructureFile/ChemBookStructure3/GIF/CB1176014.gif)
300559-61-9
13 suppliers
$189.00/500mg
Yield:300559-61-9 66%
Reaction Conditions:
with triethylamine in N,N-dimethyl-formamide at 20; for 0.5 h;
Steps:
Methyl 2-chloroacetylamino-4,5,6,7-tetrahydrobenzo[b]thiophene-3-carboxylate (4), methyl 2-chloroacetylamino-6-methyl-4,5,6,7-tetrahydrobenzo[b]thiophene-3-carboxylate (5), and methyl 6-tert-butyl-2-chloroacetylamino-4,5,6,7-tetrahydrobenzo[b]thiophene-3-carboxylate (6)
General procedure: Chloroacetyl chloride (1.1 g, 0.1 mol) was added to a solution of 2-aminothiophene 11-13 (0.01 mol) and Et3N (1.0 g, 0.01 mol) in DMF (20 mL) at 20 °C with stirring. The reaction mass was stirred for 30 min, poured into H2O (50 mL), extracted with chloroform (2×10 mL), dried over Na2SO4. Chloroform was evaporated, the residue was crystallized from a mixture hexane-chloroform, 10 : 1.
References:
Sokolov;Aksinenko, A. Yu.;Gabrel’yan;Grigoriev [Russian Chemical Bulletin,2019,vol. 68,# 8,p. 1629 - 1632][Izv. Akad. Nauk, Ser. Khim.,2019,# 8,p. 1629 - 1632,4]
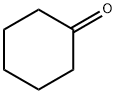
108-94-1
530 suppliers
$12.00/50g
![2-(2-CHLORO-ACETYLAMINO)-4,5,6,7-TETRAHYDRO-BENZO[B]THIOPHENE-3-CARBOXYLIC ACID METHYL ESTER](/StructureFile/ChemBookStructure3/GIF/CB1176014.gif)
300559-61-9
13 suppliers
$189.00/500mg