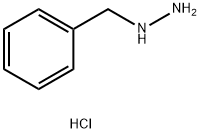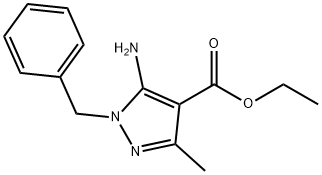
ethyl 5-amino-1-benzyl-3-methyl-1H-pyrazole-4-carboxylate synthesis
- Product Name:ethyl 5-amino-1-benzyl-3-methyl-1H-pyrazole-4-carboxylate
- CAS Number:303066-82-2
- Molecular formula:C14H17N3O2
- Molecular Weight:259.3
Yield:303066-82-2 4.45 g
Reaction Conditions:
with N-ethyl-N,N-diisopropylamine in ethanol at 90;
Steps:
1 Step 1 : Ethyl 5-amino- l-benzyl-3-methyl- lH-pyrazole-4-carboxylate
To a stirred solution of ethyl (2Z)-2-cyano-3-ethoxybut-2-enoate (5.0 g, 27.3 mmol) in ethanol (50 mL), benzylhydrazine dihydrochloride (5.3 g, 27.3 mmol) followed by N, N- diisopropylethylamine (14 mL, 81.9 mmol) were added at RT. The reaction mixture was stirred overnight at 90 °C. The ethanol was removed by evaporation and the residue obtained was basified with aqueous saturated sodium bicarbonate solution (50 mL). The aqueous mixture was extracted with ethyl acetate (100 mL x 2). The organic layer was dried over anhydrous sodium sulfate and concentrated under reduced pressure. The residue obtained was purified by silica gel column chromatography to yield 4.45 g of the titled product as a solid. lH NMR (300 MHz, DMSO- ): δ 1.25 (t, J = 6.6 Hz, 3H), 2.16 (s, 3H), 4.16 (q, J = 6.6 Hz, 2H), 5.09 (s, 2H), 6.32 (s, 2H), 7.14 (d, J = 8.1 Hz, 2H), 7.23-7.35 (m, 3H).
References:
WO2018/203298,2018,A1 Location in patent:Page/Page column 123