
CarbaMic acid, N-[2-[[(4-chlorophenyl)Methyl]aMino]ethyl]-, 1,1-diMethylethyl ester synthesis
- Product Name:CarbaMic acid, N-[2-[[(4-chlorophenyl)Methyl]aMino]ethyl]-, 1,1-diMethylethyl ester
- CAS Number:335059-94-4
- Molecular formula:C14H21ClN2O2
- Molecular Weight:284.78
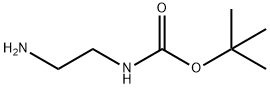
57260-73-8
464 suppliers
$5.00/5g
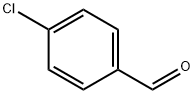
104-88-1
614 suppliers
$5.00/25g
![CarbaMic acid, N-[2-[[(4-chlorophenyl)Methyl]aMino]ethyl]-, 1,1-diMethylethyl ester](/CAS/GIF/335059-94-4.gif)
335059-94-4
13 suppliers
$262.00/250mg
Yield:335059-94-4 90%
Reaction Conditions:
with sodium tris(acetoxy)borohydride;acetic acid in 1,1-dichloroethane at 20;
Steps:
1.1
Sodium triacetoxyborohydride (3.3 g, 15.4 mmol, 1.1 eq.) was added at room temperature to a solution of 4-chlorobenzaldehyde (2 g, 14 mmol), tert-butyl 2- aminoethylcarbamate (4.5 mL, 28.5 mmol, 1.2 eq.) and acetic acid (2.5 mL) in dichloroethane (20 mL). The reaction mixture was allowed to stir overnight before being quenched with 0.5M HCl (30 mL). The mixture was then extracted with dichloromethane one time and then brine was added. The precipitate was filtered and dried to give tert-butyl 2-(4- chlorobenzylamino)ethylcarbamate (3.58 g, 90%), MS (ESI) m/e (M+H+) 285.
References:
WO2009/6569,2009,A1 Location in patent:Page/Page column 37

57260-73-8
464 suppliers
$5.00/5g
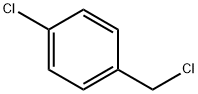
104-83-6
371 suppliers
$15.00/50g
![CarbaMic acid, N-[2-[[(4-chlorophenyl)Methyl]aMino]ethyl]-, 1,1-diMethylethyl ester](/CAS/GIF/335059-94-4.gif)
335059-94-4
13 suppliers
$262.00/250mg