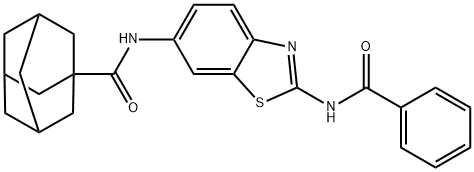
N-[2-(Benzoylamino)-6-benzothiazolyl]tricyclo[3.3.1.13,7]decane-1-carboxamide synthesis
- Product Name:N-[2-(Benzoylamino)-6-benzothiazolyl]tricyclo[3.3.1.13,7]decane-1-carboxamide
- CAS Number:362003-83-6
- Molecular formula:C25H25N3O2S
- Molecular Weight:431.55

351467-22-6
0 suppliers
inquiry
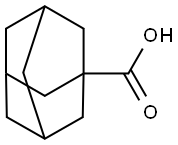
828-51-3
417 suppliers
$10.00/5g
![N-[2-(Benzoylamino)-6-benzothiazolyl]tricyclo[3.3.1.13,7]decane-1-carboxamide](/CAS/GIF/362003-83-6.gif)
362003-83-6
66 suppliers
$25.00/1mg
Yield:-
Reaction Conditions:
with 1-hydroxy-7-aza-benzotriazole;N-(3-dimethylaminopropyl)-N-ethylcarbodiimide;triethylamine in N,N-dimethyl-formamide at 60; for 2 h;
Steps:
1 Adamantane-1-carboxylic Acid (2-benzoylamino-benzothiazol-6-yl)-amide
EXAMPLE 1 Adamantane-1-carboxylic Acid (2-benzoylamino-benzothiazol-6-yl)-amide 100 mg of N-(6-amino-benzothiazol-2-yl)-benzamide, 250 mg of adamantane carboxylic acid, 24.5 mg of HOAt, 247 μl of triethylamine and 126 μl of EDC/free base) are dissolved in 2 ml of dry DMF and stirred at 60° for 2 hours. The mixture obtained is diluted with EtOAc, and extracted with 1N HCl and 5% aqueous NaHCO3 solution. From the organic layer obtained solvent is evaporated and the evaporation residue is subjected to chromatography. Adamantane-1-carboxylic acid (2-benzoylamino-benzothiazol-6-yl)-amide is obtained.
References:
Bornancin, Frederic;Oberhauser, Berndt US2009/170914, 2009, A1 Location in patent:Page/Page column 18