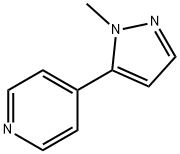
4-(1-Methyl-1H-pyrazol-5-yl)pyridine synthesis
- Product Name:4-(1-Methyl-1H-pyrazol-5-yl)pyridine
- CAS Number:905281-60-9
- Molecular formula:C9H9N3
- Molecular Weight:159.19
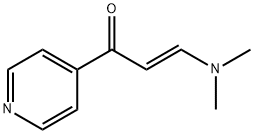
123367-27-1
50 suppliers
$55.00/250mg
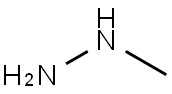
60-34-4
2 suppliers
$65.59/25g
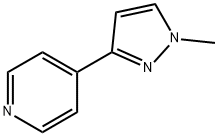
905281-61-0
0 suppliers
inquiry

905281-60-9
25 suppliers
$54.00/100mg
Yield:905281-60-9 64%
Reaction Conditions:
in ethanol; for 1 h;Heating / reflux;
Steps:
1.2
To (E)-S-(dimethylamino)-l-(pyridin-4-yl)prop-2-en- 1-one (1.0Og) in 50 mL ethanol was added 0.32 mL methylhydrazine and the resulting mixture heated to reflux for 1 hour. The solvent was removed by evaporation and partitioned between ethyl acetate and water. The aqueous layer was extracted with another portion of ethyl acetate. The combined ethyl acetate extracts were washed with brine, dried over magnesium sulfate, filtered, and evaporated to afford 0.62 g crude product as a yellow oil. The material was purified by chromatography, eluting with a 10:1 mixture of dichloromethane-methanol to yield 0.30 g (64% yield) product as a yellow oil. 1H NMR (CDCl3) was consistent with a 3.8:1 mixture of desired product and undesired isomer (4-(l -methyl- l-H-pyrazole-3- yl)pyridine). LC/MS of the mixture showed 2 peaks, each with a M+l molecular ion of m/z 160.2 by positive ESI.
References:
WO2006/84015,2006,A2 Location in patent:Page/Page column 74