4-(3-BROMO-PHENYL)-1H-IMIDAZOLE synthesis
- Product Name:4-(3-BROMO-PHENYL)-1H-IMIDAZOLE
- CAS Number:53848-00-3
- Molecular formula:C9H7BrN2
- Molecular Weight:223.07
Yield:53848-00-3 91%
Reaction Conditions:
with potassium carbonate in methanol at 25; for 0.5 h;
Steps:
General experimental procedures for the synthesis of N-unsubstituted imidazoles
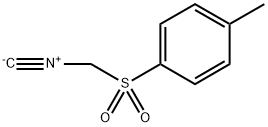
General procedure: In a round-bottom flask, to N-sulfinyl imines (1.0 mmol) and TosMIC (1.1 mmol) in MeOH (3 mL), was added K2CO3 (2.5 mmol). The suspension was allowed to stir at room temperature for 0.5 h. The progress of the reaction was monitored by TLC. After complete conversion, the reaction mixture was diluted with ethyl acetate and water. The organic layer was separated, dried over anhydrous sodium sulphate, filtered and evaporated. The crude product was subjected to column chromatography on silica gel (eluent: petroleum ether/ethyl acetate = 40:60) to provide the corresponding mono-substituted imidazoles.
References:
Babu Shubha, Priya;Prasad, Hari;Ramaiah, Manjunatha M.;Shivananju, Nanjunda Swamy [Tetrahedron Letters,2020] Location in patent:supporting information
![2-Propanesulfinamide, N-[(3-bromophenyl)methylene]-2-methyl-](/CAS/20210305/GIF/960503-83-7.gif)