
4-(4-ACETYLPIPERAZIN-1-YL)BENZALDEHYDE synthesis
- Product Name:4-(4-ACETYLPIPERAZIN-1-YL)BENZALDEHYDE
- CAS Number:890092-19-0
- Molecular formula:C13H16N2O2
- Molecular Weight:232.28
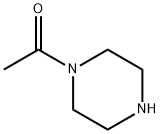
13889-98-0
313 suppliers
$5.00/5g

459-57-4
582 suppliers
$6.00/25g

890092-19-0
22 suppliers
$253.00/100mg
Yield:890092-19-0 64%
Reaction Conditions:
with potassium carbonate in N,N-dimethyl-formamide at 130; for 16 h;
Steps:
4.1 Step 1 : 4-(4-Acetylpiperazin-l-yl)benzaldehyde
Step 1 : 4-(4-Acetylpiperazin-l-yl)benzaldehyde To a solution of 4-fluorobenzaldehyde (5 g, 40.2 mmol) in N,N-dimethylformamide (130 mL) was added potassium carbonate (8.35 g, 60.4 mmol) and 1-piperazin-l-ylethanone (10.3 g, 80.5 mmol) and the reaction was stirred at 130 °C for 16 hours. The reaction was then cooled to ambient temperature, diluted with EtOAc and washed with water (x2) and brine. The organic layer was then dried with MgSC>4, concentrated and purified by silica gel column chromatography (0-10% methanol in dichloromethane) to give 4-(4- acetylpiperazin-l-yl)benzaldehyde (5.96 g, 64% yield). LCMS (m/z) ES+ 233 [M+l]+.
References:
WO2013/92941,2013,A1 Location in patent:Page/Page column 44
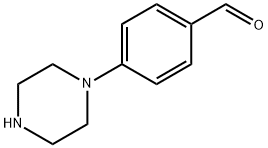
27913-98-0
55 suppliers
$45.00/50mg
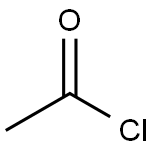
75-36-5
563 suppliers
$17.92/100G

890092-19-0
22 suppliers
$253.00/100mg
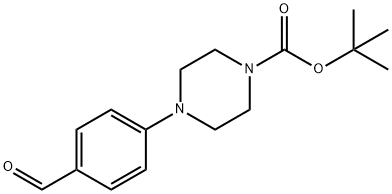
197638-83-8
104 suppliers
$7.00/250mg

890092-19-0
22 suppliers
$253.00/100mg