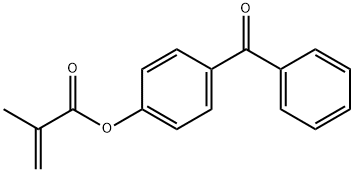
4-Benzoylphenyl Methacrylate synthesis
- Product Name:4-Benzoylphenyl Methacrylate
- CAS Number:56467-43-7
- Molecular formula:C17H14O3
- Molecular Weight:266.29

80-62-6
592 suppliers
$18.00/25mL
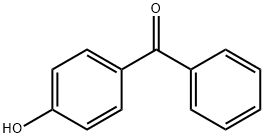
1137-42-4
492 suppliers
$6.00/25g

56467-43-7
46 suppliers
$74.00/5G
Yield: 1320.4 g
Reaction Conditions:
with methacryloyl anhydride;2,4-dimethyl-6-tert-butylphenol;sodium methylate in methanol at 60 - 90; for 6 h;Reagent/catalyst;
Steps:
1 Example 1
Example 1Sodium Methacrylate Catalyst Generated in situ10045] Apparatus: 2 1 four-necked round-bottom flask with mechanical stirring, reflux condenser, air inlet, PtiOO temperature sensor, oil bath.10046] Batch: 1.5 mol (297.9 g) of 4-hydroxybenzophe- none, 1.65 mol (270.3 g) of MAAH, 0.0087 mol (1.6 g) of NM3O solution, 399 mg of 2,4-dimethyl-6-tert-butylphenol (Topanol A);10047] Preparation of a 30% strength solution of 4-(meth- acryloyloxy)benzophenone in MMA: Addition of a total of 772 g of MMA10048] Theoretical yield of 4-(methacryloyloxy)benzo- phenone: 399 g10049] Procedure: 4-Hydroxybenzophenone was weighed out, and 126 g of the subsequently required MMA were added; a readily stirrable composition was formed. Then IVL&AH, Topanol A stabilizer and NM3O catalyst were added, air was introduced, and the batch was heated to 90° C. The course of the reaction was monitored by gas chromatography (a sample (approximately 1.5 ml) taken, diluted with approximately 5 ml of acetone, and filtered). After a total of 5 h at 90° C., the batch was cooled to 60° C., 30 g of methanol were added for conversion of the remaining MAAH, and the batch was stirred 60° C. for 1 h. The remainder of the MMA (646 g) was then added to the batch, to produce a 30% strength solution. After cooling with accompanying stirring, filtering took place through a pressure filter with Seitz Ti000 filter layer (014 cm). Yield:1320.4 g
References:
EVONIK ROEHM GmbH;KLESSE, Wolfgang;KNEBEL, Joachim;SAAL, Doris US2016/297738, 2016, A1 Location in patent:Paragraph 0045-0049;0065-0069;0079-0083;0094-0101;0111-0120

1137-42-4
492 suppliers
$6.00/25g
920-46-7
325 suppliers
$20.00/1g

56467-43-7
46 suppliers
$74.00/5G
760-93-0
305 suppliers
$27.60/50g

1137-42-4
492 suppliers
$6.00/25g

56467-43-7
46 suppliers
$74.00/5G

67-56-1
737 suppliers
$7.29/5ml-f

760-93-0
305 suppliers
$27.60/50g

1137-42-4
492 suppliers
$6.00/25g

80-62-6
592 suppliers
$18.00/25mL

56467-43-7
46 suppliers
$74.00/5G