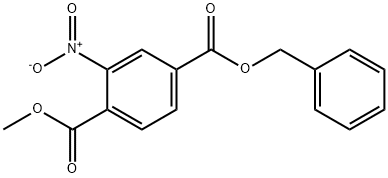
4-Benzyl 1-Methyl 2-nitroterephthalate synthesis
- Product Name:4-Benzyl 1-Methyl 2-nitroterephthalate
- CAS Number:402925-14-8
- Molecular formula:C16H13NO6
- Molecular Weight:315.28
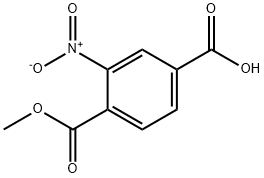
35092-89-8
125 suppliers
$42.53/5g
28547-28-6
46 suppliers
inquiry
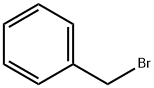
100-39-0
423 suppliers
$10.00/10g

402925-14-8
6 suppliers
inquiry
Yield:402925-14-8 63%
Reaction Conditions:
with potassium carbonate in N,N-dimethyl-formamide;
Steps:
72.B benzyl 3-nitro-4-methoxycarbonylbenzoate
Example 72B benzyl 3-nitro-4-methoxycarbonylbenzoate To a solution of methyl 2-nitro-4-carboxybenzoate (1.13 g, 5.00 mmol), K2CO3 (680 mg, 4.92 mmol) in N,N-dimethylformamide (5 mL) was added benzyl bromide (750 μL, 6.30 mmol). The mixture was heated at 100° C. for 15 minutes, cooled to ambient temperature, poured into water (30 mL) and extracted with diethyl ether (3*10 mL). The combined ether layers were washed with water (2*10 mL), brine (1*10 mL), dried (MgSO4), filtered, and concentrated under reduced pressure. The residue was crystallized from 20% ethyl acetate/hexanes to provide the titled compound (1.0 g, 63%). 1H NMR (300 MHz, d6-DMSO) δ8.52 (d, 1, J=1.7), 8.39 (dd, 1, J=1.7, 8.1), 8.01 (d, 1, J=7.8), 7.53-7.35 (m, 5), 5.43(s,2), 3.89 (s,3); MS (ESI) m/z=333 (M+NH4+).
References:
US2002/35137,2002,A1