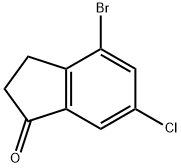
4-Bromo-6-chloro-indan-1-one synthesis
- Product Name:4-Bromo-6-chloro-indan-1-one
- CAS Number:1260017-94-4
- Molecular formula:C9H6BrClO
- Molecular Weight:245.5
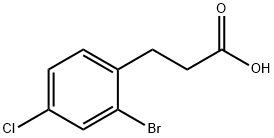
66192-04-9
12 suppliers
inquiry

1260017-94-4
24 suppliers
$45.00/10mg
Yield:1260017-94-4 3.7 g
Reaction Conditions:
with aluminum (III) chloride;oxalyl dichloride in dichloromethane at 40; for 3 h;Reflux;
Steps:
4 Step 4: 4-Bromo-6-chloro-2,3-dihydro-1 H-inden-1 -one
Oxalylchloride (3.6 ml_) is added dropwise to a solution of 3-(2-bromo-4- chlorophenyl)propanoic acid (4.4 g) in dichloromethane (40 ml_). The m ixture is stirred for 2 hours at 40°C and then treated with AICI3 (2.7 g). The mixture is heated to reflux for 1 hour, cooled to room temperature and partitioned between ethyl acetate and saturated aqueous NaHCO3 solution. The aqueous phase is extracted twice with ethyl acetate and the combined organic phases are washed with brine. After drying (Na2SO4) the solvents are evaporated. The residue is chromatographed on silica gel (cyclohexane/ethyl acetate 95:5→90:10) to give the title compound. Yield: 3.7 g; LC (method 1 ): tR = 1 .20 min; Mass spectrum (ESI+): m/z = 245 [M+H]+.
References:
WO2013/144098,2013,A1 Location in patent:Page/Page column 123
66191-92-2
2 suppliers
inquiry

1260017-94-4
24 suppliers
$45.00/10mg

2019-34-3
219 suppliers
$6.00/1g

1260017-94-4
24 suppliers
$45.00/10mg