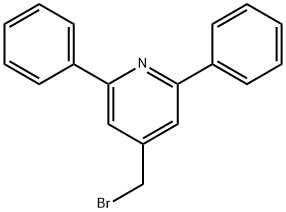
4-(bromomethyl)-2,6-diphenylpyridine synthesis
- Product Name:4-(bromomethyl)-2,6-diphenylpyridine
- CAS Number:796096-61-2
- Molecular formula:C18H14BrN
- Molecular Weight:324.21

796096-62-3
3 suppliers
inquiry

796096-61-2
7 suppliers
inquiry
Yield:796096-61-2 92%
Reaction Conditions:
with dibromotriphenylphosphorane in dichloromethane at 20; for 4 h;
Steps:
24.2
A solution of ethyl-2, 6-DIPHENYL-ISONICOTINATE (930 mg, 3.06 mmol) in anhydrous tetrahydrofuran (10 mL) was added dropwise to a stirred slurry of lithium aluminium hydride (116 mg, 3.06 mmol) in tetrahydrofuran (20 mL) at 0°C. The reaction was stirred at 0°C for 1 hour and then quenched by the addition of water (0.12 mL), 2N sodium hydroxide (0.12 mL) and finally water (0.36 mL). Celite was added and the reaction was diluted with diethyl ether (50 mL), stirred for 10 mins, and filtered. The ethereal solution was dried over anhydrous MGS04, filtered and concentrated in vacuo to yield the primary alcohol as a white solid (782 mg, 98 %). Dibromotriphenylphosphorane (1.40 g, 3.24 mmol) was added as a solid to a solution of the alcohol (prepared in the previous reduction step) (775 mg, 2.95 mmol) in anhydrous DICHLOROMETHABE (40 mL). The reaction was stirred at room temperature for 4 hours and then partitioned between water and diethyl ether. The phases were separated, the aqueous phase being further extracted with diethyl ether (3 x 30 mL). The combined extract was washed with water and brine. The ethereal solution was dried over anhydrous MGS04, filtered and concentrated in vacuo to yield the title compound as a white solid (874 mg, 92 %) ; Rf : 0.8 (30% ethyl acetate in heptane); 1H NMR (CDCl3, 300 MHz) δ 8.16 (2H, s, ArH), 8.12 (2H, s, ArH), 7.68 (2H, s, ArH), 7.48 (6H, m, ArH), 4.52 (2H, s, CH2Br).
References:
WO2004/99192,2004,A2 Location in patent:Page 82-83