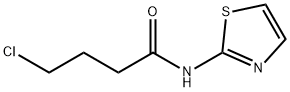
4-CHLORO-N-(1,3-THIAZOL-2-YL)BUTANAMIDE synthesis
- Product Name:4-CHLORO-N-(1,3-THIAZOL-2-YL)BUTANAMIDE
- CAS Number:37762-99-5
- Molecular formula:C7H9ClN2OS
- Molecular Weight:204.68

96-50-4
513 suppliers
$5.00/5g
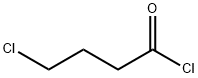
4635-59-0
468 suppliers
$17.67/10gm:

37762-99-5
16 suppliers
$45.00/10mg
Yield:37762-99-5 92%
Reaction Conditions:
with potassium carbonate in toluene at 20; for 24 h;
Steps:
7.1.1. 4-Chloro-N-(thiazol-2-yl)-butanamide 7
A mixture of 2-aminothiazole (4, 4.0 g, 0.04 mol), 4-chlorobutyryl chloride (6, 11.3 g, 0.08 mol), and potassium carbonate (1.4 g, 0.01 mol) in toluene (100 ml) was stirred at room temperature for 24 h. The toluene was then evaporated under reduced pressure. The residue was then quenched with water, stirred, and filtered. The solid obtained was washed, dried and recrystallized from water to give the required product 7; 92% yield, mp 125-127 °C, 1H NMR (CDCl3): δ 12.92 (s, 1H, NH), 7.54 (d, 1H, J = 3.5 Hz, thiazole-H), 7.04 (d, 1H, J = 3.5 Hz, thiazole-H), 3.71 (t, 2H, J = 6.0 Hz, CH2), 2.81 (t, 2H, J = 6.0 Hz, CH2), 2.31-2.26 (m, 2H, CH2). 13C NMR: δ 27.3, 32.8, 44.4, 113.6, 136.2, 160.1, 170.0. MS: m/z 113 (100%), M = 204 (20%), M + 2 = 206 (5%). (C7H9ClN2OS) C, H, N.
References:
El-Subbagh, Hussein I.;Hassan, Ghada S.;El-Azab, Adel S.;Abdel-Aziz, Alaa A.-M.;Kadi, Adnan A.;Al-Obaid, Abdulrahman M.;Al-Shabanah, Othman A.;Sayed-Ahmed, Mohamed M. [European Journal of Medicinal Chemistry,2011,vol. 46,# 11,p. 5567 - 5572] Location in patent:experimental part