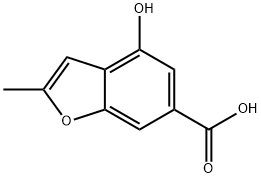
4-hydroxy-2-methylbenzofuran-6-carboxylicacid synthesis
- Product Name:4-hydroxy-2-methylbenzofuran-6-carboxylicacid
- CAS Number:37978-62-4
- Molecular formula:C10H8O4
- Molecular Weight:192.17
314725-17-2
42 suppliers
$297.60/1g

37978-62-4
7 suppliers
inquiry
Yield:37978-62-4 96%
Reaction Conditions:
with water;sodium hydroxide in tetrahydrofuran;methanol at 20; for 12 h;
Steps:
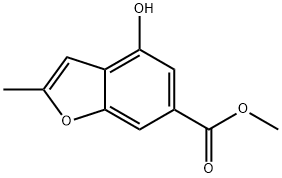
18.2 Step 2) Synthesis of 4-hydroxy-2-methylbenzofuran-6-carboxylic acid
Methyl 4-hydroxy-2-methylbenzofuran-6-carboxylate (2.06 g, 10 mmol), methanol (20 mL), tetrahydrofuran (20 mL) and water (20 mL) were gradually added to a 250 mL single neck flask, then sodium hydroxide (2.0 g, 50 mmol) was added. The reaction mixture was stirred for 12 h at rt. The mixture was concentrated in vacuo to remove solvent. To the residue was added water (120 mL). The mixture was washed with ether (80 mL), and the aqueous phase was acidified to pH 1 with 2 N dilute hydrochloric acid and extracted with ethyl acetate (80 mL x 2). The combined organic phases were washed with saturated brine (80 mL x 2), dried over anhydrous sodium sulfate, filtered, and concentrated in vacuo to give the title compound as a white solid(1.84 g, 96%).MS (ES-API, neg. ion) m/z: 191.0 [M - 1]t
References:
WO2017/36404,2017,A1 Location in patent:Page/Page column 99

338454-02-7
55 suppliers
$53.00/100mg

37978-62-4
7 suppliers
inquiry