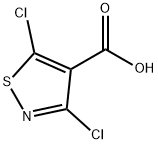
4-Isothiazolecarboxylic acid, 3,5-dichloro synthesis
- Product Name:4-Isothiazolecarboxylic acid, 3,5-dichloro
- CAS Number:3889-59-6
- Molecular formula:C4HCl2NO2S
- Molecular Weight:198.03
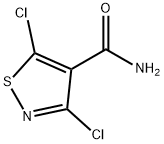
3925-01-7
0 suppliers
inquiry

3889-59-6
19 suppliers
inquiry
Yield:3889-59-6 75%
Reaction Conditions:
Stage #1: 3,5-dichloroisothiazole-4-carboxamidewith sulfuric acid at 100; for 2 h;
Stage #2: with sodium nitrite in water at 50;Concentration;Temperature;
Steps:
151.1 Step 1; Dichloro-1, 2-thiazole-4-carboxylic acid
Into a 500-mL round-bottom flask, was placed dichloro-1, 2-thiazole-4- carbonitrile (36 g, 201.09 mmol, 1.00 equiv), Cone, sulfuric acid (100 mL).The resulting solution was stirred for 2 h at 100°C in an oil bath. This was followed by the addition of a solution of NaN02 (15 g, 217.39 mmol, 1.08 equiv) in water (100 mL) dropwise with stirring at 0°C. The resulting solution was allowed to react, with stirring, for overnight while the temperature was maintained at 50°C in an oil bath. The resulting solution was diluted with 500 mL of water. The resulting solution was extracted with 5x500 mL of ethyl acetate and the organic layers combined. The resulting mixture was washed with 1x500 mL of brine. The mixture was dried over anhydrous sodium sulfate and concentrated under vacuum. This resulted in 30 g (75%) of dichloro- 1 ,2-thiazole-4-carboxylic acid as red oil. Mass spectrum (ESI, m/z): Calcd. for C4HCI2NO2S, 197.1 (M+H), found 197.1.
References:
WO2015/134039,2015,A1 Location in patent:Page/Page column 176-177
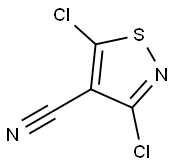
2120-82-3
51 suppliers
inquiry

3889-59-6
19 suppliers
inquiry