
4-(T-BOC-AMINOMETHYL)-3-CHLOROBENZOIC ACID synthesis
- Product Name:4-(T-BOC-AMINOMETHYL)-3-CHLOROBENZOIC ACID
- CAS Number:165528-69-8
- Molecular formula:C13H16ClNO4
- Molecular Weight:285.72

24424-99-5
825 suppliers
$13.50/25G
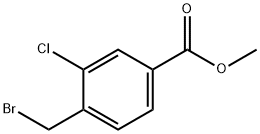
74733-30-5
79 suppliers
$17.00/250mg

165528-69-8
8 suppliers
inquiry
Yield:165528-69-8 67%
Reaction Conditions:
Stage #1: methyl 4-Bromomethyl-3-chlorobenzoatewith ammonia in ethanol at 20; for 1 h;
Stage #2: with diethyl ether in ethanol;
Stage #3: di-tert-butyl dicarbonatewith sodium hydroxide in 1,4-dioxane;water at 20; for 18 h;
Steps:
4.B 4B. 4-(tert-Butyloxycarbonylaminomethyl)-3-chlorobenzoic acid
To a saturated solution of ammonia in ethanol (170 ml) was added methyl 4-bromomethyl-3-chlorobenzoate from Example 4A (5.5 g, 20.9 mmol). The mixture was stirred at room temperature for 1 h and then concentrated in vacuo. The residue was triturated with diethyl ether and the resultant white crystals were filtered off and washed with more diethyl ether. To a solution of this solid in water (100 ml) were added solutions of di-tert-butyl dicarbonate (5.0 g, 23.0 mmol) in dioxan (100 ml) and sodium hydroxide (1.86 g, 46.0 mmol) in water (100 ml). The mixture was stirred at room temperature for 18h and then concentrated in vacuo. The aqueous residue was acidified with citric acid and extracted with chloroform/2-propanol. The organic layer was washed with water, dried over MgSO4, and concentrated in vacuo to give a white solid identified as 4-(tert-butyloxycarbonylaminomethyl)-3-chlorobenzoic acid (2.8 g, 67%).
References:
EP1449844,2004,A1 Location in patent:Page 20

24424-99-5
825 suppliers
$13.50/25G

940062-11-3
27 suppliers
$45.00/10mg

165528-69-8
8 suppliers
inquiry
56525-63-4
139 suppliers
$15.00/5g

165528-69-8
8 suppliers
inquiry

74733-30-5
79 suppliers
$17.00/250mg

165528-69-8
8 suppliers
inquiry