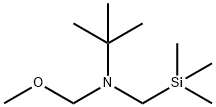
N-(Methoxymethyl)-2-methyl-N-[(trimethylsilyl)-methyl]-2-propanamine synthesis
- Product Name:N-(Methoxymethyl)-2-methyl-N-[(trimethylsilyl)-methyl]-2-propanamine
- CAS Number:455954-96-8
- Molecular formula:C10H25NOSi
- Molecular Weight:203.4

67-56-1
774 suppliers
$9.00/25ml
50-00-0
885 suppliers
$10.00/25g
![2-Methyl-N-[(trimethylsilyl)methyl]-2-propanamine](/CAS/GIF/79250-80-9.gif)
79250-80-9
10 suppliers
$60.00/1g
![N-(Methoxymethyl)-2-methyl-N-[(trimethylsilyl)-methyl]-2-propanamine](/CAS/GIF/455954-96-8.gif)
455954-96-8
8 suppliers
inquiry
Yield:-
Reaction Conditions:
Stage #1: methanol;formaldehyd;N-tert-butyltrimethylsilylmethylamine in water at 0; for 2.75 h;
Stage #2: with potassium carbonate in water at 0; for 0.5 h;
Steps:
2
2-Methyl-N-[(trimethylsilyl)methyl]propan-2-amine (from preparation 1) (4.31 g, 27 mmol) was added to an ice-cooled mixture of methanol (1.29 mL, 31.8 mmol) and aqueous formaldehyde (37% w/v 2.49 mL, 33 mmol) over 45 minutes. The heterogeneous mixture was stirred at 0° C. for 2 hours and then solid potassium carbonate (325 mesh) (1.08 g, 13 mmol) was added and the mixture was stirred for 30 minutes at 0° C. The layers were separated and the aqueous phase was extracted with ethyl acetate (3×20 mL). The combined organic layers were dried over sodium sulfate, filtered, and evaporated under reduced pressure to give an 80:20 mixture of the title compound and unreacted tert-butyl[(trimethylsilyl)methyl]amine as a colourless oil (5.09 g). The mixture was used directly without further purification. 1H NMR (CD3OD, 400 MHz) δ 0.04 (s, 9H), 1.11 (s, 9H), 2.27 (s, 2H), 3.34 (s, 3H), 4.17 (s, 2H).
References:
US2008/234280,2008,A1 Location in patent:Page/Page column 28