
5-BROMO-N-TERT-BUTYLNICOTINAMIDE synthesis
- Product Name:5-BROMO-N-TERT-BUTYLNICOTINAMIDE
- CAS Number:342013-78-9
- Molecular formula:C10H13BrN2O
- Molecular Weight:257.13
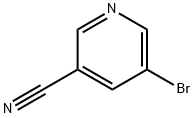
35590-37-5
226 suppliers
$6.00/250mg
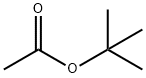
540-88-5
351 suppliers
$22.00/25mL

342013-78-9
29 suppliers
$51.00/1/ G
Yield:342013-78-9 93%
Reaction Conditions:
Stage #1: 5-bromopyridine-3-carbonitrile;acetic acid tert-butyl esterwith sulfuric acid at 20 - 45;
Stage #2: with sodium hydrogencarbonate in water;
Steps:
5
Method 5To a stirred suspension of 5-bromo-3-cyanopyridine (366 mg, 2.0 mmol) in tert-butyl acetate (2.25 mL) was added concentrated sulphuric acid (0.15 mL) dropwise at room temperature. The reaction mixture was heated at 45 C and stirred overnight. After this time the reaction mixture was poured onto saturated NaHCO3 (20 mL) and extracted with ethyl acetate (40 mL). The organic extracts were concentrated in vacuo to afford the title compound; no further purification was required (480 mg, 1.87 mmol, 93%).
References:
WO2009/68482,2009,A1 Location in patent:Page/Page column 47
20826-04-4
540 suppliers
$5.00/1g

342013-78-9
29 suppliers
$51.00/1/ G
39620-02-5
69 suppliers
$82.00/10g
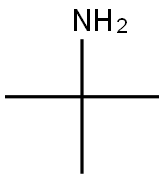
75-64-9
419 suppliers
$10.00/10g

342013-78-9
29 suppliers
$51.00/1/ G