
5-Hexenamide synthesis
- Product Name:5-Hexenamide
- CAS Number:28487-09-4
- Molecular formula:C6H11NO
- Molecular Weight:113.16
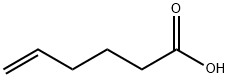
1577-22-6
130 suppliers
$13.00/250mg

28487-09-4
0 suppliers
inquiry
Yield:28487-09-4 97%
Reaction Conditions:
Stage #1: 5-hexenoic acidwith 4-methyl-morpholine;isobutyl chloroformate in tetrahydrofuran at 0; for 0.5 h;
Stage #2: with ammonium hydroxide in tetrahydrofuran at 20;
Steps:
Hex-5-enamide S58.
To a solution of 5-hexenoic acid (0.44 mL, 3.701 mmol) dissolved in THF (5 mL) was added N-methylmorpholine (0.45 mL, 4.07 1 mmol) and the solution was cooled to 0°C. Isobutyl chloroformate (0.53 mL, 4.07 1 mmol) wasadded dropwise and the reaction was stirred at 0°C for 30 minutes, then ammonium hydroxide (28% NH3 in H20, 0.64 mL) was added and the reaction was allowed to warm to room temperature and stir overnight. The reaction was quenched with sat. NH4C1 and extracted with EtOAc 3x. The combined organic layers were washed with 1M HC1 and brine, dried over MgSO4, filtered and concentrated, yielding the titlecompound as a white solid (407 mg, 97% yield). 1H NMR (400 MHz, CDC13) ? 5.79(ddt, J = 17.0, 10.2, 6.7 Hz, 1H), 5.34 (br s, 2H), 5.10-4.95 (m, 2H), 2.28-2.21 (m,1H), 2.12 (dd, J = 14.2, 7.1 Hz, 2H), 1.82- 1.70 (m, 1H); 13C NMR (101 MHz,CDC13) ? 175.73, 137.90, 115.51, 35.16, 33.16, 24.57; IR (film) 3361 (brN-H), 3184(brN-H), 2944, 2359, 2342, 1633 (C=O), 1415, 1229, 1135, 1077, 991, 908, 775,667; HRMS Accurate mass (ES): Found 114.0917 (-1.8 ppm), C6H12NO (M+H)requires 114.0919; MP 70.0- 75.1°C.
References:
WO2017/184558,2017,A1 Location in patent:Page/Page column 109; 110

5048-19-1
94 suppliers
$110.00/1g

28487-09-4
0 suppliers
inquiry
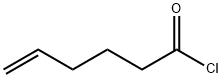
36394-07-7
10 suppliers
inquiry

28487-09-4
0 suppliers
inquiry