
5-IODO-2-ISOPROPOXY-PYRIDINE synthesis
- Product Name:5-IODO-2-ISOPROPOXY-PYRIDINE
- CAS Number:902837-54-1
- Molecular formula:C8H10INO
- Molecular Weight:263.08
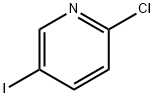
69045-79-0
351 suppliers
$5.00/1g
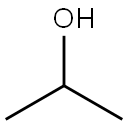
67-63-0
1550 suppliers
$17.00/25ML

902837-54-1
19 suppliers
$125.00/1g
Yield:902837-54-1 95%
Reaction Conditions:
Stage #1: isopropyl alcoholwith sodium hydride in N,N-dimethyl-formamide at 0 - 20;Inert atmosphere;
Stage #2: 2-choro-5-iodopyridine in N,N-dimethyl-formamide at 60;Inert atmosphere;
Steps:
3.1. General Procedure for the Nucleophilic Substitution on 3-Iodopyridine Derivatives
General procedure: The 0.4 g (10 mmol) of 60% NaH was dissolved in 10 mL dry DMF under an N2 atmosphere.The suspension was cooled to 0 C, and 10 mmol of the appropriate alcohol was added. The solutionwas allowed to warm up to room temperature. When the formation of H2 gas stopped, the solutionof 5 mmol of the appropriate 3-iodopyridine (1,3,5) in 15 mL DMF was added, and the mixture wasstirred at ambient temperature (for 1) or at 60 C (for 3 and 5) until no further conversion was observed.The mixture was then diluted with water and extracted with EtOAc. The combined organic layerswere dried over Na2SO4, filtered, and concentrated under reduced pressure. The crude product waspurified via preparative reversed-phase chromatography using 25 mM aqueous NH4HCO3 solutionand acetonitrile as eluents or via flash chromatography on silica gel using DCM, MeOH, heptane,and EtOAc as eluents.
References:
Farkas, Emese;Gyorfi, Nandor;Kotschy, Andras;Nemet, Norbert;Novak, Zoltan;Weber, Csaba [Molecules,2020,vol. 25,# 20,art. no. 4766]