
3-(4-FLUORO-PHENYL)-4,5-DIHYDRO-ISOXAZOLE-5-CARBOXYLIC ACID synthesis
- Product Name:3-(4-FLUORO-PHENYL)-4,5-DIHYDRO-ISOXAZOLE-5-CARBOXYLIC ACID
- CAS Number:522615-28-7
- Molecular formula:C10H8FNO3
- Molecular Weight:209.17
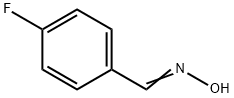
459-23-4
50 suppliers
$13.43/250mgs:
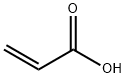
79-10-7
689 suppliers
$20.50/500ml

522615-28-7
21 suppliers
$90.00/50mg
Yield:522615-28-7 73%
Reaction Conditions:
with pyridine;N-chloro-succinimide in chloroform at 20;Inert atmosphere;Cooling with ice;
Steps:
4.3. General synthetic procedure for aryl-isoxazoline acid 3a-c
General procedure: A solution of the corresponding aryl-aldoxime (2 mmol) in CHCl3 (2 mL) was added dropwise to an ice-cooled solution of CHCl3 (8 mL), acrylic acid (2.3 mmol), pyridine (3 mmol) and N-chlorosuccinimide (NCS) (0.31 g, 2.3 mmol) under nitrogen for 30 min. The reaction was then stirred at room temperature for several hours and monitored to the completion by thin-layer chromatography. The solution was washed with water and dried with Na2SO4. The filtrate was evaporated under a reduced pressure and the residue purified by recrystallization from appropriate solvents. The physico-chemical properties and spectral data for representative compounds are as follows
References:
Shi, Liqiao;Hu, Rui;Wei, Yanhong;Liang, Ying;Yang, Ziwen;Ke, Shaoyong [European Journal of Medicinal Chemistry,2012,vol. 54,p. 549 - 556] Location in patent:experimental part
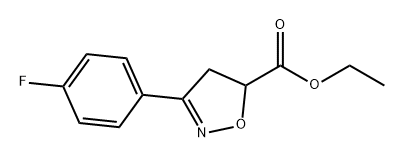
1382998-36-8
0 suppliers
inquiry

522615-28-7
21 suppliers
$90.00/50mg