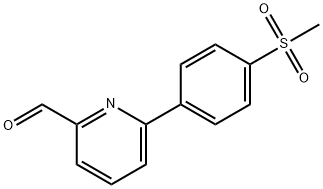
6-(4-(METHYLSULFONYL)PHENYL)-2-PYRIDINE& synthesis
- Product Name:6-(4-(METHYLSULFONYL)PHENYL)-2-PYRIDINE&
- CAS Number:834884-84-3
- Molecular formula:C13H11NO3S
- Molecular Weight:261.3
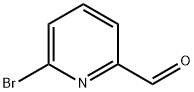
34160-40-2
336 suppliers
$6.00/1g
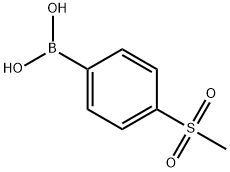
149104-88-1
254 suppliers
$15.00/1g

834884-84-3
18 suppliers
$92.59/1g
Yield:834884-84-3 95%
Reaction Conditions:
with tetrakis(triphenylphosphine) palladium(0);potassium carbonate in ethanol;water at 80; for 5 h;Inert atmosphere;
Steps:
7 Preparation of 6-(4-(Methylsulfonyl)phenyl)picolinaldehyde (3)
A solution of 6-bromopicolinaldehyde (1, 2.00 g, 10.8 mmol), (4-(methylsulfonyl)phenyl)boronic acid (2, 2.36 g, 11.8 mmol), and potassium carbonate (2.22 g, 16.2 mmol) in ethanol/ water (200/20 mL) under nitrogen was treated with tetrakis(triphenylphosphine)palladium(0) (0.62 g, 0.54 mmol). The mixture was heated at 80° C. for 5 h. The reaction mixture was cooled to room temperature and concentrated. The residue was purified by silica gel chromatography eluting with) -50% EtOAc in hexanes to afford the title compound (2.66 g, 95%) as a yellow solid: 1H NMR (300 MHz, CDCl3) δ 10.18 (s, 1H), 8.31 (d, J=8.5 Hz, 2H), 8.10 (d, J=8.5 Hz, 2H), 7.97-7.07 (m, 3H), 3.11 (s, 3H); Multimode MS m/z 262 [M+H]+
References:
US2014/140956,2014,A1 Location in patent:Paragraph 0673; 0674