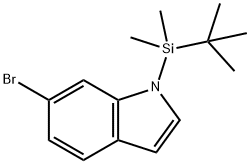
6-Bromo-1-(tert-butyldimethylsilyl)indole synthesis
- Product Name:6-Bromo-1-(tert-butyldimethylsilyl)indole
- CAS Number:184637-11-4
- Molecular formula:C14H20BrNSi
- Molecular Weight:310.3
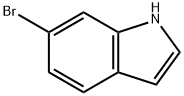
52415-29-9
421 suppliers
$9.00/1g
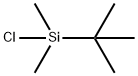
18162-48-6
668 suppliers
$9.00/5g

184637-11-4
14 suppliers
$45.00/100mg
Yield:184637-11-4 100%
Reaction Conditions:
Stage #1: 6-bromo-1H-indolewith sodium hydride in N,N-dimethyl-formamide;mineral oil at 0 - 20; for 0.5 h;
Stage #2: tert-butyldimethylsilyl chloride in N,N-dimethyl-formamide;mineral oil at 0 - 20; for 1 h;
Steps:
2.2. General procedure B for the synthesis of N-alkylindoles
General procedure: Sodium hydride (60% in mineral oil, 1.10 equiv) was suspended in DMF [0.6 M]. 1H-Indole (1.00 equiv)was solubilized in DMF [1.0 M] and added to the suspension at 0 °C. The mixture was stirred at rt for 30min. Bromoalkyl or TMSCl (1.50 equiv) was diluted in DMF [3.0 M] and added to the solution at 0 °C.The mixture was stirred at rt for 1 hour. The solution was quenched with water (20 mL) and extractedthree times with EtOAc (10 mL). The organic layers were combined, dried over MgSO4 and concentratedunder reduced pressure. The liquid was filtered through a 5 cm pad of silica with 100% pentane or Et2O toafforded the title compound.
References:
Grenet, Erwann;Das, Ashis;Caramenti, Paola;Waser, Jér?me [Beilstein Journal of Organic Chemistry,2018,vol. 14,p. 1208 - 1214] Location in patent:supporting information