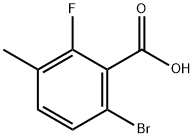
6-Bromo-2-fluoro-3-methylbenzoic acid synthesis
- Product Name:6-Bromo-2-fluoro-3-methylbenzoic acid
- CAS Number:743466-98-0
- Molecular formula:C8H6BrFO2
- Molecular Weight:233.03
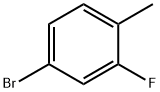
51436-99-8
327 suppliers
$5.00/1g
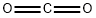
124-38-9
122 suppliers
$175.00/23402

743466-98-0
24 suppliers
inquiry
Yield: 86.2%
Reaction Conditions:
Stage #1:2-fluoro-4-bromotoluene with lithium diisopropyl amide in tetrahydrofuran at -78; for 1 h;
Stage #2:carbon dioxide in tetrahydrofuran at 20;
Steps:
1.39.1 Step 1: Preparation of methyl 6-bromo-2-fluoro-3-methylbenzoic acid
Step 1:
Preparation of methyl 6-bromo-2-fluoro-3-methylbenzoic acid
To a solution of 4-bromo-2-fluoro-1-methylbenzene (21.9 g, 116 mmol) in THF (100 mL) was added lithium diisopropylamide (LDA) (2 M solution in THF, 69 mL, 139 mmol) at -78° C.
After being stirred at -78° C. for 1 h, the reaction was treated with excess solid carbon dioxide, and stirred while the reaction temperature slowly rose to room temperature.
The mixture was concentrated and then partitioned between 4N NaOH and ethyl acetate.
The aqueous phase was adjusted to pH 2 with 2N HCl and then extracted three times with ethyl acetate.
The combined extracts were washed with water and brine, dried over with anhydrous Na2SO4, filtered and concentrated under reduced pressure.
The residue was purified by column chromatography on silica gel eluted with EA to give methyl 6-bromo-2-fluoro-3-methylbenzoic acid (23.3 g, 86.2%) as a white solid.
References:
Anacor Pharmaceuticals, Inc.;Eli Lilly and Company;Akama, Tsutomu;Balko, Terry William;Defauw, Jean Marie;Plattner, Jacob J.;White, William Hunter;Winkle, Joseph Raymond;Zhang, Yong-Kang;Zhou, Yasheen US2013/131016, 2013, A1 Location in patent:Paragraph 0401; 0402