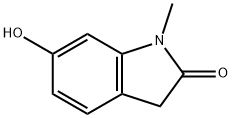
6-HYDROXY-1-METHYLINDOLIN-2-ONE synthesis
- Product Name:6-HYDROXY-1-METHYLINDOLIN-2-ONE
- CAS Number:13383-73-8
- Molecular formula:C9H9NO2
- Molecular Weight:163.17

7703-91-5
12 suppliers
inquiry

13383-73-8
25 suppliers
inquiry
Yield:13383-73-8 66.61%
Reaction Conditions:
with BBr3 in dichloromethane at 0 - 20; for 0.5 h;Inert atmosphere;
Steps:
6.2 Step 2: Synthesis of Compound 6-2
At room temperature, compound 6-1 (0.75 g) was added to a two-necked flask, DCM (10 mL) was added, cooled in an ice bath, protected by N2, and then BBr3 (4.23 g, 1.57 mL) was added dropwise. After the drop was completed, the reaction was carried out in an ice bath. 30min. Under ice bath, add ice water to the reaction solution to quench the reaction, adjust pH with saturated NaHCO, separate the organic phase, extract the aqueous phase with EA, wash the EA layer with water, wash with saturated brine, and concentrate to obtain a light brown solid product 6-2 (0.46 g, 66.61% yield).
References:
CN113979995,2022,A Location in patent:Paragraph 0100-0101; 0104-0105
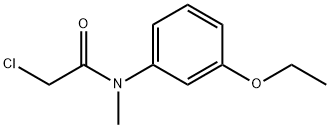
861069-79-6
0 suppliers
inquiry

13383-73-8
25 suppliers
inquiry
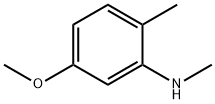
156267-11-7
0 suppliers
inquiry

13383-73-8
25 suppliers
inquiry
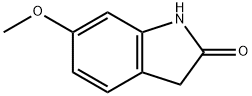
7699-19-6
106 suppliers
$60.00/100mg

13383-73-8
25 suppliers
inquiry
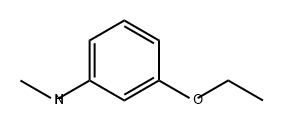
91240-01-6
0 suppliers
inquiry

13383-73-8
25 suppliers
inquiry