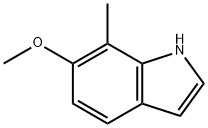
6-METHOXY-7-METHYLINDOLE synthesis
- Product Name:6-METHOXY-7-METHYLINDOLE
- CAS Number:19500-05-1
- Molecular formula:C10H11NO
- Molecular Weight:161.2
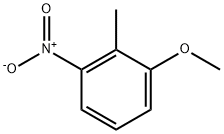
4837-88-1
238 suppliers
$12.72/1gm:
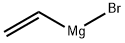
1826-67-1
210 suppliers
$60.89/100ml

19500-05-1
31 suppliers
$45.00/1mg
Yield:19500-05-1 27%
Reaction Conditions:
in tetrahydrofuran at -40; for 0.5 h;
Steps:
6-Methoxy-7-methyl- 1 H-indolc:To a solution of l-methoxy-2-methyl~3~nitro-benzene (5.00 g, 29.9 mmol) in THF ( 100 mL) was added vinyl magnesium bromide (89.9 mL, 89.9 mmol) in one portion at -40 C. The mixture was stirred at -40 C for 30 min, was quenched by pouring into aqueous ammonium chloride saturated solution. The aqueous solution was extracted with EtOAc. The combined organic phases were dried over MgSO4 and concentrated in vacuo. The crude oil was purified by column chromatography (hexanes to 20% EtOAc in hexanes gradient) to give 6-methoxy- 7-methyl- l H-indoie (1.32 g, 27%) as brown solid. 1H NMR (400 MHz, CDCh) ?: 7.94 (br, IH), 7.46 (m, IH), 7.16 (m, IH), 6.89 (m, IH), 6.54 (m, IH), 3.94 (s, 3H), 2.40 (s, 3H); ESMS mA- 162 (M+H)+
References:
WO2009/120655,2009,A1 Location in patent:Page/Page column 38