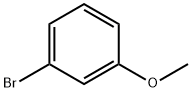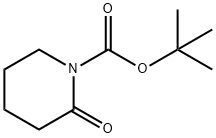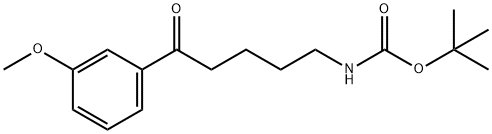
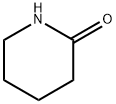
Carbamic acid, N-[5-(3-methoxyphenyl)-5-oxopentyl]-, 1,1-dimethylethyl ester synthesis
- Product Name:Carbamic acid, N-[5-(3-methoxyphenyl)-5-oxopentyl]-, 1,1-dimethylethyl ester
- CAS Number:616226-78-9
- Molecular formula:C17H25NO4
- Molecular Weight:307.38
675-20-7
353 suppliers
$5.00/1g

24424-99-5
820 suppliers
$13.50/25G
36282-40-3
91 suppliers
$112.00/100ml
![Carbamic acid, N-[5-(3-methoxyphenyl)-5-oxopentyl]-, 1,1-dimethylethyl ester](/CAS/20210111/GIF/616226-78-9.gif)
616226-78-9
2 suppliers
inquiry
Yield:616226-78-9 92%
Reaction Conditions:
Stage #1: piperidin-2-onewith n-butyllithium in tetrahydrofuran;hexane at -78;Inert atmosphere;
Stage #2: di-tert-butyl dicarbonate in tetrahydrofuran;hexane at -78; for 3 h;Inert atmosphere;
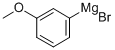
Stage #3: (3-methoxyphenyl)magnesium bromide in tetrahydrofuran;hexane at -78; for 3 h;Inert atmosphere;
Steps:
1 7.1.2.1. Tert-Butyl 5-(3-methoxyphenyl)-5-oxopentylcarbamate (20)
To a solution of δ-valerolactam (1 g, 10.10 mmol) in anhydrous THF (27 mL) at -78 °C under an argon atmosphere was added dropwise 2.5 M butyllithium in hexanes (4.04 mL, 10.09 mmol) and after 30 min of stirring, a solution of di-tert-butyldicarbonate (2.2 g, 10.10 mmol) in anhydrous THF (3.5 mL) was added and temperature was maintained at -78 °C for 3 h. Then a solution of 3-methoxyphenylmagnesium bromide 16, prepared from 2.45 g of 3-bromoanisole and 368 mg of magnesium turnings in anhydrous THF (14 mL), was added and stirring was continued at the same temperature for further 3 h. The reaction was allowed to warm to room temperature and quenched with 2.0 M HCl (10 mL). The mixture was extracted with diethyl ether (3 * 15 mL) and the combined organic layers were washed with a 5% NaHCO3 aqueous solution (1 * 20 mL) and brine (1 * 20 mL), dried over anhydrous sodium sulphate and concentrated in vacuo to obtain 20 as a yellow oil (2.84 g, 92% yield). Rf = 0.21 (cyclohexane/ethyl acetate 9:1). 1H NMR: δ 1.43 (s, 9H), 1.51-1.61 (m, 2H), 1.70-1.81 (m, 2H), 2.97 (t, J = 7.2 Hz, 2H), 3.14 (t, J = 6.4 Hz, 2H), 3.84 (s, 3H), 4.63 (br s, 1H), 7.09 (ddd, J = 8.3, 2.8, 1.1 Hz, 1H), 7.35 (t, J = 8.3 Hz, 1H), 7.46-7.47 (m, 1H), 7.50-7.53 (m, 1H). 13C NMR: δ 21.2, 28.4 (3C), 29.4, 38.4, 40.0, 55.8, 79.5, 115.9, 118.7, 122.1, 129.3, 138.9, 155.9, 160.5, 185.1. MS (ESI) m/z [M+H]+ Calcd for C17H26NO4+: 308.19. Found: 308.2.
References:
Matera, Carlo;Quadri, Marta;Sciaccaluga, Miriam;Pomè, Diego Yuri;Fasoli, Francesca;De Amici, Marco;Fucile, Sergio;Gotti, Cecilia;Dallanoce, Clelia;Grazioso, Giovanni [European Journal of Medicinal Chemistry,2016,vol. 108,p. 392 - 405]

675-20-7
353 suppliers
$5.00/1g
![Carbamic acid, N-[5-(3-methoxyphenyl)-5-oxopentyl]-, 1,1-dimethylethyl ester](/CAS/20210111/GIF/616226-78-9.gif)
616226-78-9
2 suppliers
inquiry

24424-99-5
820 suppliers
$13.50/25G
![Carbamic acid, N-[5-(3-methoxyphenyl)-5-oxopentyl]-, 1,1-dimethylethyl ester](/CAS/20210111/GIF/616226-78-9.gif)
616226-78-9
2 suppliers
inquiry