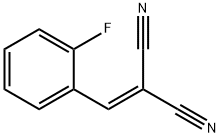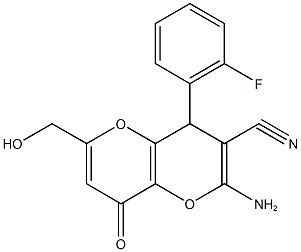
2-amino-4-(2-fluorophenyl)-6-(hydroxymethyl)-8-oxo-4,8-dihydropyrano[3,2-b]pyran-3-carbonitrile synthesis
- Product Name:2-amino-4-(2-fluorophenyl)-6-(hydroxymethyl)-8-oxo-4,8-dihydropyrano[3,2-b]pyran-3-carbonitrile
- CAS Number:665000-66-8
- Molecular formula:C16H11FN2O4
- Molecular Weight:314.2679

501-30-4
663 suppliers
$5.00/25mg

446-52-6
431 suppliers
$6.00/25g

109-77-3
442 suppliers
$10.00/5g
![2-amino-4-(2-fluorophenyl)-6-(hydroxymethyl)-8-oxo-4,8-dihydropyrano[3,2-b]pyran-3-carbonitrile](/CAS/20180711/GIF/665000-66-8.gif)
665000-66-8
7 suppliers
inquiry
Yield:665000-66-8 85%
Reaction Conditions:
in water at 50; for 0.333333 h;Sonication;
Steps:
2.2. Typical procedure for synthesis of 2-amino-4,8-dihydropyrano[3,2-b]pyran-3-carbonitriles (4a) under ultrasound irradiation
General procedure: A 25 mL Erlenmeyer flask was charged with malononitrile (66 mg, 1 mmol), benzaldehyde (1 mmol), and kojic acid (1 mmol) and water (5 mL). The reaction flask was located in the ultrasonic bath, where the surface of reactants is slightly lower than the level of the water, and irradiated under 20%, 40%, 60%, 80% and 100% of the power of the ultrasonic bath at 50 C (bath temperature, the temperature inside the reactor was also 50 C) for the period of time (The reaction was monitored by TLC) separately as indicated in Table 3. The reaction temperature was controlled by addition or removal of water from ultrasonic bath. After completion of the reaction, the mixture was diluted with water (10 mL), the solid was filtered, washed with water and dried to give crude product, which was further purified by recrystallization from ethanol to offer pure product 4a in 95% yield.
References:
Banitaba, Sayed Hossein;Safari, Javad;Khalili, Shiva Dehghan [Ultrasonics Sonochemistry,2013,vol. 20,# 1,p. 401 - 407] Location in patent:supporting information