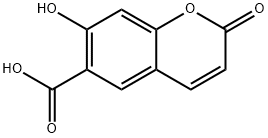
7-Hydroxycoumarin-6-carboxylic acid synthesis
- Product Name:7-Hydroxycoumarin-6-carboxylic acid
- CAS Number:833-52-3
- Molecular formula:C10H6O5
- Molecular Weight:206.15

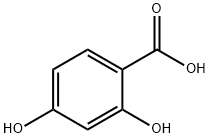
52591-14-7
0 suppliers
inquiry

833-52-3
31 suppliers
inquiry
Yield:833-52-3 55%
Reaction Conditions:
with sodium hydroxide in 1,4-dioxane;water; for 0.333333 h;Reflux;
Steps:
7-Hydroxy-2-oxo-2H-chromene-6-carboxylic acid (2).
Peuruthenicin (1), 1.0 g (4.5 mmol), was dis-solved in 200 mL of dioxane, 400 mL of 20% aqueous sodium hydroxide was added, and the mixture was refluxed for 20 min. The resulting solution was cooled to 10°C, acidified to pH 3-4 by adding 70 mL of 10% sulfuric acid, and extracted with chloroform (3 × 15 mL). The combined extracts were dried over MgSO 4 , filtered, and evaporated, and the residue was recrystallized from ethanol and dried under reduced pressure. Yield 0.514 g (55%). mp 248-250°C (decomp.); published data [15]: mp 258°C (decomp.). IR spectrum, ν, cm -1 : 3504, 3394, 3066, 2849, 2702, 2472, 1705, 1678, 1628, 1597, 1579, 1392, 1294, 1242, 1147, 1119, 916, 862, 810, 760, 727, 633. UV spectrum (EtOH), λ max , nm (log ε): 208 (3.69), 224 (3.58), 238 (3.56), 305 (3.32), 330 (3.50). 1 H NMR spectrum, δ, ppm: 6.14 d (1H, 4-H, J = 9.4 Hz), 6.70 s (1H, 8-H), 7.59 d (1H, 3-H, J = 9.4 Hz), 7.96 s (1H, 5-H), 11.25 s (OH). 13 C NMR spectrum, δ C , ppm: 95.83 (C 4a ), 103.27 (C 8 ), 110.97 (C 6 ), 111.74 (C 3 ), 131.30 (C 4 ), 144.52 (C 5 ), 157.37 (C 8a ), 161.79 (C 7 ), 164.51 (C 2 ), 173.13 (C=O).
References:
Zakharov;Lipeeva;Gatilov, Yu. V.;Makarov;Shults [Russian Journal of Organic Chemistry,2019,vol. 55,# 10,p. 1518 - 1526][Zh. Org. Khim.,2019,vol. 55,# 10,p. 1567 - 1576,10]