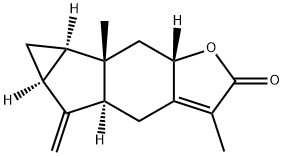
(4aS)-4aα,5,5aα,6,6aα,6b,7,7aβ-Octahydro-3,6bβ-dimethyl-5-methylenecycloprop[2,3]indeno[5,6-b]furan-2(4H)-one synthesis
- Product Name:(4aS)-4aα,5,5aα,6,6aα,6b,7,7aβ-Octahydro-3,6bβ-dimethyl-5-methylenecycloprop[2,3]indeno[5,6-b]furan-2(4H)-one
- CAS Number:70578-36-8
- Molecular formula:C15H18O2
- Molecular Weight:230.3
![Methyl (2E)-2-[(1aR,1bS,5aS,6aS)-octahydro-1b-methyl-6-methylenecycloprop[a]inden-4(1H)-ylidene]propanoate](/CAS/20210305/GIF/1369537-08-5.gif)
1369537-08-5
0 suppliers
inquiry
![(4aS)-4aα,5,5aα,6,6aα,6b,7,7aβ-Octahydro-3,6bβ-dimethyl-5-methylenecycloprop[2,3]indeno[5,6-b]furan-2(4H)-one](/CAS/20180808/GIF/70578-36-8.gif)
70578-36-8
3 suppliers
inquiry
Yield:70578-36-8 37%
Reaction Conditions:
with 3,5-dimethyl-1H-pyrazole;chromium(VI) oxide in dichloromethane at 0; for 13 h;Inert atmosphere;Reflux;
Steps:
4.8 (+)-Sarcandralactone A (1)
To a cooled (0 °C) solution of alkene 10 (20 mg, 0.081 mmol) in DCM (10 mL) were successively added CrO3 (41 mg, 0.41 mmol) and 3,5-dimethylpyrazole (78 mg, 0.82 mmol). After being stirred at 0 °C for another 1 h, to the mixture was added Celite and heated to reflux for 12 h with an oil bath. After cooling to room temperature, the mixture was filtered through a silica gel pad and washed with Et2O. The filtrate was concentrated under reduced pressure. The residue was purified by flash chromatography (Hexane/EtOAc=150:1 to 50:1 to 20:1) to afford lactone 12 (7 mg, 37%) and alkene 10 (9 mg, 45%).
References:
Qian, Shan;Zhao, Gang [Tetrahedron,2013,vol. 69,# 52,p. 11169 - 11173]