
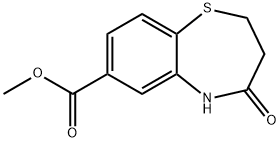
4-OXO-2,3,4,5-TETRAHYDRO-1,5-BENZOTHIAZEPINE-7-CARBOXYLIC ACID synthesis
- Product Name:4-OXO-2,3,4,5-TETRAHYDRO-1,5-BENZOTHIAZEPINE-7-CARBOXYLIC ACID
- CAS Number:790681-57-1
- Molecular formula:C10H9NO3S
- Molecular Weight:223.25
724790-46-9
0 suppliers
inquiry

790681-57-1
14 suppliers
inquiry
Yield:790681-57-1 68%
Reaction Conditions:
Stage #1: methyl 4-oxo-2,3,4,5-tetrahydro-1,5-benzothiazepine-7-carboxylatewith sodium acetate;lithium iodide in N,N-dimethyl-formamide at 150; for 4 h;
Stage #2: with hydrogenchloride in water;N,N-dimethyl-formamide at 0;
Steps:
18.d
To a solution of methyl 4-oxo-2,3,4,5-tetrahydro-1 ,5-benzothiazepine-7- carboxylate (500 mg, 2.11 mmol) in DMF (5 ml_) were added lithium iodide (1.19g, 8.90 mmol) and anhydrous sodium acetate (145 mg, 1.77 mmol). After heating at 150° for 4h, the reaction mixture was diluted with ice water. The solution was acidified with 6N HCI, followed by extraction with DCM. The organic layers were combined, washed with aqueous sodium chloride and dried over MgSθ4. Trituration with acetonitrile gave the title compound as a tan solid (317mg, 68%): LC/MS (ES) m/z 224.2 (M+H)+
References:
WO2007/16610,2007,A2 Location in patent:Page/Page column 43