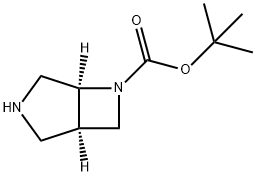
3,6-Diazabicyclo[3.2.0]heptane-6-carboxylic acid, 1,1-diMethylethyl ester, (1R,5S)- synthesis
- Product Name:3,6-Diazabicyclo[3.2.0]heptane-6-carboxylic acid, 1,1-diMethylethyl ester, (1R,5S)-
- CAS Number:799279-81-5
- Molecular formula:C10H18N2O2
- Molecular Weight:198.26
![3-benzyl 6-tert-butyl 3,6-diazabicyclo[3.2.0]heptane-3,6-dicarboxylate](/CAS/20180629/GIF/799279-84-8.gif)
799279-84-8
2 suppliers
inquiry
![3,6-Diazabicyclo[3.2.0]heptane-6-carboxylic acid, 1,1-diMethylethyl ester, (1R,5S)-](/CAS/20150408/GIF/799279-81-5.gif)
799279-81-5
42 suppliers
$239.00/250mg
Yield:799279-81-5 96%
Reaction Conditions:
with hydrogen;palladium 10% on activated carbon in methanol at 20; for 10 h;
Steps:
8.B tert-Butyl(1R,5S)-3,6-diazabicyclo[3.2.0]heptane-6-carboxylate
Example 8B tert-Butyl(1R,5S)-3,6-diazabicyclo[3.2.0]heptane-6-carboxylate The product of Example 8A (40.0 g, 0.120 mol) was dissolved in methanol (400 mL) and treated with Pd/C (10 wt %, 4.0 g) under H2 at ambient temperature for 10 h. The reaction was monitored with HPLC. After the reaction was complete, the catalyst was removed by filtration through a short column of diatomaceous earth. The filtrate was concentrated under reduced pressure to provide the title compound as a colorless oil (22.8 g, 11.5 mmol, 96% yield). 1H NMR (MeOH-d4, 300 MHz) δ 1.43 (s, 9H), 2.47 (dd, J=112.6, 3.8 Hz, 1H), 2.62 (dd, J=112.2, 5.7 Hz, 1H), 2.96 (m, 1H), 3.05 (d, J=112.2 Hz, 1H), 3.22 (d, J=112.5 Hz, 1H), 3.45 (m, 1H), 3.95 (m, 1H), 4.63 (dd, J=6.1, 3.7 Hz, 1H); MS (DCl/NH3) m/z 199 (M+H)+. HPLC conditions: Zorbax-XDB-C8 column 4.6*250 mm with solvents H2O (0.2 v.% HClO4)/MeCN (from v.80:20 to 10:90 within 15 min.) at 1.0 mL/Min., UV detection ? 220 nm. 20/80, flow rate, 1.0 mL/min; uv 220 nm; tR=8.6 minutes.
References:
US2005/101602,2005,A1 Location in patent:Page/Page column 30-31
![(S,S)-3-CBZ-3,6-DIAZABICYCLO[3.2.0]HEPTANE](/CAS/GIF/370881-43-9.gif)
370881-43-9
25 suppliers
$285.00/100mg
![3,6-Diazabicyclo[3.2.0]heptane-6-carboxylic acid, 1,1-diMethylethyl ester, (1R,5S)-](/CAS/20150408/GIF/799279-81-5.gif)
799279-81-5
42 suppliers
$239.00/250mg
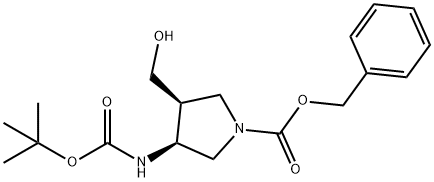
246510-69-0
9 suppliers
inquiry
![3,6-Diazabicyclo[3.2.0]heptane-6-carboxylic acid, 1,1-diMethylethyl ester, (1R,5S)-](/CAS/20150408/GIF/799279-81-5.gif)
799279-81-5
42 suppliers
$239.00/250mg
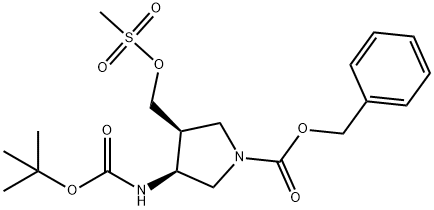
246510-70-3
2 suppliers
inquiry
![3,6-Diazabicyclo[3.2.0]heptane-6-carboxylic acid, 1,1-diMethylethyl ester, (1R,5S)-](/CAS/20150408/GIF/799279-81-5.gif)
799279-81-5
42 suppliers
$239.00/250mg
![1-Pyrrolidinecarboxylic acid, 3-amino-4-[[(methylsulfonyl)oxy]methyl]-, phenylmethyl ester, (3S,4S)-, mono(trifluoroacetate) (9CI)](/CAS/20211123/GIF/799279-83-7.gif)
799279-83-7
1 suppliers
inquiry
![3,6-Diazabicyclo[3.2.0]heptane-6-carboxylic acid, 1,1-diMethylethyl ester, (1R,5S)-](/CAS/20150408/GIF/799279-81-5.gif)
799279-81-5
42 suppliers
$239.00/250mg