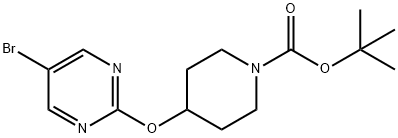
4-(5-Bromopyrimidin-2-yloxy)piperidine-1-carboxylic acid tert-butyl synthesis
- Product Name:4-(5-Bromopyrimidin-2-yloxy)piperidine-1-carboxylic acid tert-butyl
- CAS Number:832735-41-8
- Molecular formula:C14H20BrN3O3
- Molecular Weight:358.23
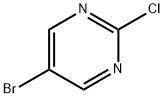
32779-36-5
622 suppliers
$5.00/5g
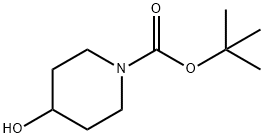
109384-19-2
469 suppliers
$5.00/1g

832735-41-8
15 suppliers
inquiry
Yield:832735-41-8 48%
Reaction Conditions:
with caesium carbonate in N,N-dimethyl-formamide at 20; for 14 h;
Steps:
1.1 1)
1) Production of 2-(1-t-butoxycarbonylpiperidin-4-yloxy)-5-bromopyrimidine: 1-t-butoxycarbonyl-4-hydroxypiperidine (408 mg, 2.03 mmol) and cesium carbonate (764 mg, 2.34 mmol) were added to a DMF solution (10 ml) of 2-chloro-5-bromopyrimidine (300 mg, 1.56 mmol), and stirred at room temperature for 14 hours. Water was added to the reaction mixture, and extracted with ethyl acetate. The organic layer was washed with saturated saline, dried with anhydrous sodium sulfate, and concentrated under reduced pressure. The residue was purified through silica gel column chromatography (C-300, hexane:ethyl acetate = 10:1) to obtain the entitled compound (268 mg, 48 %).
References:
EP1642898,2006,A1 Location in patent:Page/Page column 54