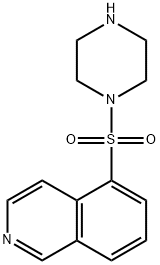
1-(5-ISOQUINOLINESULFONYL)PIPERAZINE, DIHYDROCHLORIDE synthesis
- Product Name:1-(5-ISOQUINOLINESULFONYL)PIPERAZINE, DIHYDROCHLORIDE
- CAS Number:84468-24-6
- Molecular formula:C13H15N3O2S
- Molecular Weight:277.34
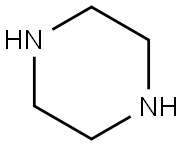
110-85-0
552 suppliers
$5.00/5 g

84468-15-5
104 suppliers
$178.00/250mg

84468-24-6
88 suppliers
$72.00/5mg
Yield: 89%
Reaction Conditions:
with potassium carbonate in dichloromethane
Steps:
3 EXAMPLE 3
EXAMPLE 3 In 100 ml of methylene chloride were added 6.0 g of piperazine and 1.2 g of anhydrous potassium carbonate, and to the mixture was added dropwise 30 ml of a methylene chloride solution containing 2.0 g of 5-isoquinolinesulfonyl chloride under cooling with ice. After the dropwise addition of the methylene chloride solution, the mixed solution was stirred at a temperature of 15° C. to 25° C. for 15 hours, and then the reaction solution was washed with water, dried with anhydrous magnesium sulfate, and the methylene chloride was distilled therefrom. The residue thus obtained was subjected to a silica gel column chromatography (silica gel: 70 g; solvent: chloroform) to give 2.14 g of 1-(5-isoquinolinesulfonyl)piperazine, i.e., Compound (35) in a yield of 89%. Mass spectrum (m/e): 277, 234, 212, 191 and 128 NMR spectrum (CDCl3): 1.65(1H, NH), 2.8-3.3(8H, 4*NCH2), 7.5-7.9(1H), 8.2-8.7(4H) and 9.35(1H) IR absorption spectrum (νmaxKBr, cm-1): 3350, 1600, 1560, 1540, 1370 and 1160
References:
Asahi Kasei Kogyo Kabushiki Kaisha;Hidaka;Hiroyoshi US4456757, 1984, A
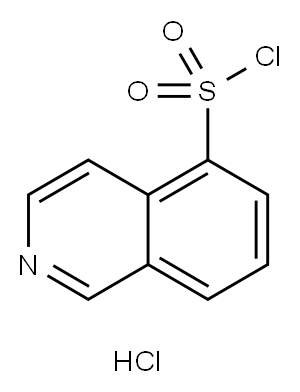
105627-79-0
141 suppliers
$20.00/100mg
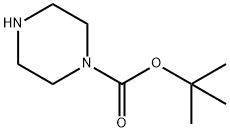
57260-71-6
740 suppliers
$5.00/5g

84468-24-6
88 suppliers
$72.00/5mg