
Tert-butyl [(1r,2s)-2-ethenyl-1-{[(1-Methylcyclopropyl)sulfonyl]carbaMoyl}cyclopropyl]carbaMate synthesis
- Product Name:Tert-butyl [(1r,2s)-2-ethenyl-1-{[(1-Methylcyclopropyl)sulfonyl]carbaMoyl}cyclopropyl]carbaMate
- CAS Number:853269-57-5
- Molecular formula:C15H24N2O5S
- Molecular Weight:344.43
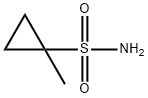
669008-26-8
152 suppliers
$9.00/100mg
159622-10-3
146 suppliers
$12.00/250mg
![Tert-butyl [(1r,2s)-2-ethenyl-1-{[(1-Methylcyclopropyl)sulfonyl]carbaMoyl}cyclopropyl]carbaMate](/CAS/20150408/GIF/853269-57-5.gif)
853269-57-5
16 suppliers
inquiry
Yield:853269-57-5 79%
Reaction Conditions:
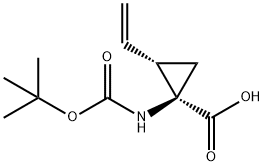
Stage #1: (1R,2S)-1-(tert-butoxycarbonylamino)-2-vinylcyclopropanecarboxylic acidwith 1,1'-carbonyldiimidazole in tetrahydrofuran at 85; for 0.5 h;
Stage #2: 1-methylcyclopropane-1-sulfonamidewith 1,8-diazabicyclo[5.4.0]undec-7-ene in tetrahydrofuran at 20; for 16 h;
Steps:
C.216.1
To a solution of lR-tert-butoxycarbonylamino-2S-vinyl-cyclopropanecarboxylic acid (2.3 g, 10.1 mmol) in THF (40 mL) was added CDI (1.80 g, 11.6 mmol) and was heated to 85 °C for 30 min. After let cool to rt, the reaction mixture was treated with 1-methyl-cyclopropanesulfonam (1.64 g, 12.1 mmol) and DBU (3.08 g, 20.2 mmol). After stirring at rt for 16 h, the reaction was diluted with EtOAc (160 mL) and washed with 2x25 mL IN HC1. The combined aqueous layer was extracted with 1x50 mL EtOAc. The combined organic layer was washed with H20 (50 mL), brine, dried over MgS04 and concentrated to a light yellow solid product (2.75 g, 79%). The product was used as crude. MS m/z 367 (M+Na).
References:
WO2005/51410,2005,A1 Location in patent:Page/Page column 126

159622-10-3
146 suppliers
$12.00/250mg
![Tert-butyl [(1r,2s)-2-ethenyl-1-{[(1-Methylcyclopropyl)sulfonyl]carbaMoyl}cyclopropyl]carbaMate](/CAS/20150408/GIF/853269-57-5.gif)
853269-57-5
16 suppliers
inquiry
![Cyclopropanecarboxylic acid, 1-[[(1,1-dimethylethoxy)carbonyl]amino]-2-ethenyl-, (1R,2S)-, methyl ester, 4-methylbenzenesulfonate (1:1)](/CAS/20210305/GIF/1265883-97-3.gif)
1265883-97-3
0 suppliers
inquiry
![Tert-butyl [(1r,2s)-2-ethenyl-1-{[(1-Methylcyclopropyl)sulfonyl]carbaMoyl}cyclopropyl]carbaMate](/CAS/20150408/GIF/853269-57-5.gif)
853269-57-5
16 suppliers
inquiry
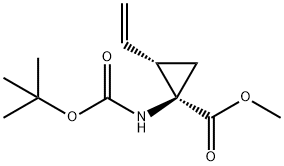
159622-09-0
33 suppliers
inquiry
![Tert-butyl [(1r,2s)-2-ethenyl-1-{[(1-Methylcyclopropyl)sulfonyl]carbaMoyl}cyclopropyl]carbaMate](/CAS/20150408/GIF/853269-57-5.gif)
853269-57-5
16 suppliers
inquiry
![Cyclopropanecarboxylic acid, 1-[[(1,1-dimethylethoxy)carbonyl]amino]-2-ethenyl-, ethyl ester, (1R,2S)-](/CAS/GIF/259217-95-3.gif)
259217-95-3
78 suppliers
$13.00/250mg
![Tert-butyl [(1r,2s)-2-ethenyl-1-{[(1-Methylcyclopropyl)sulfonyl]carbaMoyl}cyclopropyl]carbaMate](/CAS/20150408/GIF/853269-57-5.gif)
853269-57-5
16 suppliers
inquiry