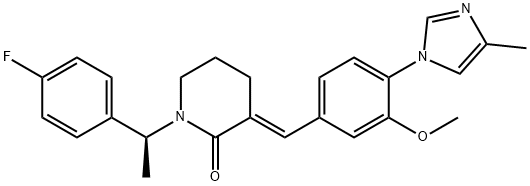
(E)-1-[(1S)-1-(4-Fluorophenyl)ethyl]-3-[3-methoxy-4-(4-methyl-1H-imidazol-1-yl)benzylidene]piperidin-2-one synthesis
- Product Name:(E)-1-[(1S)-1-(4-Fluorophenyl)ethyl]-3-[3-methoxy-4-(4-methyl-1H-imidazol-1-yl)benzylidene]piperidin-2-one
- CAS Number:870843-42-8
- Molecular formula:C25H26FN3O2
- Molecular Weight:419.49
![Ethanamine, N-[(3,5-dichlorophenyl)methylene]-2,2-diethoxy-](/CAS/20210305/GIF/1000210-73-0.gif)
1000210-73-0
0 suppliers
inquiry
![(E)-1-[(1S)-1-(4-Fluorophenyl)ethyl]-3-[3-methoxy-4-(4-methyl-1H-imidazol-1-yl)benzylidene]piperidin-2-one](/CAS/GIF/870843-42-8.gif)
870843-42-8
76 suppliers
$77.00/5mg
Yield: 77%
Reaction Conditions:
Stage #1:1-[(S)-1-(4-fluorophenyl)ethyl]-3-{hydroxy[3-methoxy-4-(4-methylimidazol-1-yl)phenyl]methyl}piperidin-2-one with p-toluenesulfonylanhydride;sodium tosylate in toluene at 100; for 36 h;
Stage #2: with sodium hydroxide;water in tetrahydrofuran;toluene at 20; for 0.05 h;Product distribution / selectivity;
Steps:
21
Example 21 Synthesis of (3E)-1-[(1S)-1-(4-fluorophenyl)ethyl]-3-[3-methoxy-4-(4-methyl-1H-imidazol-1-yl)benzylidene]piperidin-2-one p-Toluenesulfonic anhydride (194 mg, 0.59 mmol) and sodium p-toluenesulfonate (89 mg, 0.46 mmol) were sequentially added to 1-[1-(4-fluorophenyl)ethyl]-3-{hydroxy[3-methoxy-4-(4-methylimidazol-1-yl)phenyl]methyl}piperidin-2-one (200 mg, 0.46 mmol). Then, toluene (4.0 mL) was added and the mixture was stirred at 100°C for 36 hours. Water (4 mL), toluene (4 mL), a sodium hydroxide solution (2 N, 4 mL) and tetrahydrofuran (6 mL) were sequentially added to the reaction mixture. After stirring at room temperature for three minutes, toluene (4 mL) and tetrahydrofuran (2 mL) were added. A sodium hydroxide solution (2 N, 3 mL) was added to the separated organic layer which was then filtered through a celite pad. Then, toluene (6 mL) was added to the filtrate and the layers were separated. The organic layer was quantitatively determined by HPLC analysis to provide 147 mg (yield: 77%) of the title compound.
References:
Eisai R&D Management Co., Ltd. EP1950211, 2008, A1 Location in patent:Page/Page column 64