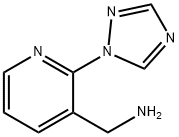
1-[2-(1H-1,2,4-triazol-1-yl)-3-pyridinyl]methanamine(SALTDATA: FREE) synthesis
- Product Name:1-[2-(1H-1,2,4-triazol-1-yl)-3-pyridinyl]methanamine(SALTDATA: FREE)
- CAS Number:950769-01-4
- Molecular formula:C8H9N5
- Molecular Weight:175.19
![2-[1,2,4]triazol-1-yl-nicotinonitrile](/CAS/GIF/950769-00-3.gif)
950769-00-3
4 suppliers
inquiry
![1-[2-(1H-1,2,4-triazol-1-yl)-3-pyridinyl]methanamine(SALTDATA: FREE)](/CAS/20200515/GIF/950769-01-4.gif)
950769-01-4
11 suppliers
$84.00/250mg
Yield:950769-01-4 92%
Reaction Conditions:
with ammonia;hydrogen;Raney 2800 nickel in methanol at 20; for 11 h;
Steps:
10.B
A catalytic amount of Raney 2800 nickel in water (Aldrich; withdrawn as a wet slurry with the tip of a spatula) was added under air to a translucent slurry of 2-[1,2,4]triazol-1-yl-nicotinonitrile (1.053 g, 6.16 mmol), as prepared in the previous step, in 2 M NH3/MeOH (50 mL). The flask was then sealed with a septum, the air was removed from the system, and the flask was flushed with H2 gas under balloon pressure. The reaction was stirred at rt under H2 for 11 h (TLC showed no starting material remained), and the bulk of the Raney nickel was then removed with a magnet. The translucent white reaction mixture was then diluted with DCM (50 mL), filtered, and the translucent filtrate was concentrated under vacuum to provide the title compound as a beige crystalline solid (988 mg, 92%).
References:
US2007/225282,2007,A1 Location in patent:Page/Page column 36-37