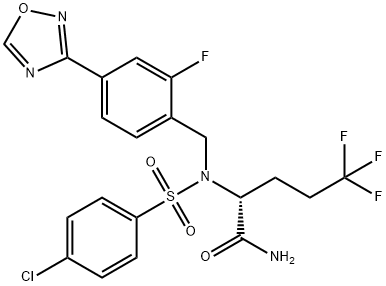
Avagacestat (BMS-708163) synthesis
- Product Name:Avagacestat (BMS-708163)
- CAS Number:1146699-66-2
- Molecular formula:C20H17ClF4N4O4S
- Molecular Weight:520.89
![Pentanamide, 2-[[(4-chlorophenyl)sulfonyl][[2-fluoro-4-[(hydroxyamino)iminomethyl]phenyl]methyl]amino]-5,5,5-trifluoro-, (2R)-](/CAS/20210305/GIF/1146699-70-8.gif)
1146699-70-8
122-51-0

1146699-66-2
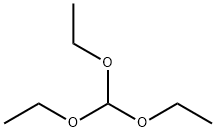
Step C. (R)-2-(4-chloro-N-(2-fluoro-4-(N'-hydroxyformamidinyl)benzyl)-benzenesulfonylamino)-5,5,5-trifluoropentanamide (246 g) was added to the reactor, followed by the addition of dry acetonitrile (509 mL), triethyl orthoformate (120 mL) and trifluoroacetic acid (7 mL). The reaction mixture was heated to 40-50 °C and the progress of the reaction was monitored by HPLC until the relative area percentage (AP) of the starting material was below 0.15. Upon completion of the reaction, a one-time addition of methanol (1.48 L) followed by water (1.034 L) was added to maintain the reaction system temperature at 45-50 °C. The reaction mixture was then cooled to 15-20 °C and the solid was collected by filtration. The filter cake was washed with a solvent mixture of acetonitrile:methanol:water (2:6:5, v/v/v) and dried under vacuum at 50-60 °C to afford (2R)-2-[N-[(4-chlorophenyl)sulfonyl]-N-[2-fluoro-4-(1 ,2,4-oxadiazol-3-yl)benzyl]amino]-5,5,5-trifluoropentanamide as a white solid (228 g, 90% yield) . 1H NMR (CDCl3, 300MHz) δ: 1.40-1.58 (m, 1H), 1.75-1.90 (m, 1H), 1.92-2.07 (m, 1H), 2.10-2.26 (m, 1H), 4.37 (dd, J = 8.67, 6.22Hz, 1H), 4.48 (d, J = 15.64Hz, 1H ), 4.64 (d, J = 15.82 Hz, 1H), 5.54 (s, 1H), 6.33 (s, 1H), 7.44-7.54 (m, 2H), 7.62 (t, J = 7.72 Hz, 1H), 7.68-7.76 (m, 3H), 7.85 (dd, J = 7.91, 1.51 Hz, 1H), 8.76 (s, 1H). 1H). 13C NMR (DMSO-d6, 75MHz) δ: 170.34, 167.75, 165.80, 159.64 (d, J = 244.5Hz, 1C), 138.19, 137.64, 131.25 (d, J = 3.75Hz, 1C), 129.31, 129.23, 129.05 (d, J = 14.25 Hz, 1C), 126.74 (q, J = 274.5 Hz, 1C), 126.91, 126.80, 123.12 (d, J = 3.75 Hz, 1C), 113.7 (d, J = 24.0 Hz, 1C), 57.92, 41.38 (d, J = 4.5 Hz, 1C), 30.04 (d, J = 30.0Hz, 1C), 22.90. 19F NMR (CDCl3, 282MHz) δ: -116.3, -66.5. IR (KBr): 3454, 334, 3286, 2952, 1705, 1432, 1325, 1260, 1167, 1084, 828 cm-1. Elemental analysis: Calculated values C20H17ClF4N4O4S: C, 46.11; H, 3.29; N, 10.71; S, 6.15; F, 14.58; Cl, 6.80. Measured values: C, 46.06; H, 3.24; N, 10.71; S, 6.25; F, 14.60; Cl, 6.88.
![1,2,4-Oxadiazole, 3-[4-(broMoMethyl)-3-fluorophenyl]-](/CAS/GIF/1146699-64-0.gif)
1146699-64-0
31 suppliers
$190.00/100mg
![PentanaMide, 2-[[(4-chlorophenyl)sulfonyl]aMino]-5,5,5-trifluoro-, (2R)-](/CAS/GIF/1146699-67-3.gif)
1146699-67-3
14 suppliers
inquiry

1146699-66-2
121 suppliers
$35.00/1mg
Yield:1146699-66-2 50%
Reaction Conditions:
with hydroxylamine;tetra-(n-butyl)ammonium iodide;caesium carbonate in acetonitrile at 15 - 35;Product distribution / selectivity;
Steps:
3.B.B.2
Step B, Procedure 2 To a suitable vessel was added (R)-2-(4-chlorophenylsulfonamido)-5,5,5-trifluoropentanamide (2.68 kg, 7.77 mol, 1 eq), 3-(4-(bromomethyl)-3-fluorophenyl)-1,2,4-oxadiazole (2.00 kg, 7.78 mol, 1 eq), cesium carbonate (1.65 kg, 5.06 mol, 0.65 eq), tetrabutylammonium iodide (0.29 kg, 0.78 mol, 0.1 eq) and acetonitrile (12.0 L 4.5 L/kg). The reaction was heated to 35° C. until complete by HPLC (3-(4-(bromomethyl)-3-fluorophenyl)-1,2,4-oxadiazole <0.5 relative AP by HPLC). The reaction was cooled to 15° C. and water (10.72 L, 4 L/kg) was added with stirring followed by glacial acetic acid (0.22 kg) to bring the pH of the reaction to <6.5. The stirring was stopped and the phases were separated (the top layer contained the product). To the product rich layer was added toluene (26.8 kg, 31 L, 10 kg/kg) followed by brine solution (20% w/w, 6.39 kg, 2 L/kg) and the layers were separated (the top layer contained the product). The mixture was distilled at ~50° C. under vacuum (200 mbar) until acetonitrile was removed. The concentration was adjusted with additional toluene if needed after distillation to ensure total volume in the reactor was ~10 L/kg. Isopropyl alcohol (0.48 kg, 0.2 L/kg) was charged and the batch was cooled to 15° C. to initiate crystallization. The resulting slurry was filtered and washed with cold toluene (18.65 kg, 21.56 L, 8 L/kg). The crude cake was tray dried under vacuum at 50° C. until loss on drying was <1.0%. The dry cake was added to a 100 L reactor along with isopropyl alcohol (27.34 kg, 34.8 L, 13 L/kg) and hydroxylamine (50% aqueous solution, 0.05 kg, 1.51 mol, 0.2 eq). The mixture was heated to 65° C. and monitored by HPLC until (R)-2-(4-chloro-N-(4-cyano-2-fluorobenzyl)phenylsulfonamido)-5,5,5-trifluoropentanamide was <0.4 AP. The reaction was then distilled (pot temperature ~50° C., vacuum 300 mbar) until reaction volume was ~60% of original. Acetonitrile (5.36 kg, 2 L/kg) was charged and the reaction temperature was increased to 70° C. to achieve complete dissolution. Water (11.26 L, 4.2 L/kg) was charged slowly while keeping the reaction temperature >65° C. The reaction was cooled to 15° C. over 2 hours and crystallization occurred. The slurry was filtered and washed with cold aqueous isopropyl alcohol (2:1 IPA:water by volume). The cake was dried in a vacuum oven until loss on drying was <1%. The product was then recrystallized by dissolving in acetonitrile (2 L/kg based on weight of input of dried cake) and methanol (6 L/kg) and then heated to 50° C. Water (4 L/kg) was added slowly, keeping the reaction temperature >50° C. The reaction was cooled to 15° C. over 2 hours. The resulting slurry was filtered and washed with a solution of methanol:acetonitrile:water (6:2:4, 5 L/kg) to give (2R)-2-[[(4-chlorophenyl)sulfonyl][[2-fluoro-4-(1,2,4-oxadiazol-3-yl)phenyl]methyl]amino]-5,5,5-trifluoropentanamide (2.02 kg, 50% yield) as a white solid.
References:
Bristol-Myers Squibb Company US2009/111858, 2009, A1 Location in patent:Page/Page column 21-22