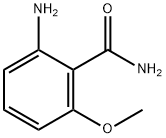
Benzamide, 2-amino-6-methoxy- synthesis
- Product Name:Benzamide, 2-amino-6-methoxy-
- CAS Number:1591-38-4
- Molecular formula:C8H10N2O2
- Molecular Weight:166.18

53600-33-2
210 suppliers
$10.00/1g

1591-38-4
18 suppliers
inquiry
Yield:1591-38-4 98%
Reaction Conditions:
Stage #1: 6-methoxyanthranilic acidwith benzotriazol-1-ol;1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride in tetrahydrofuran at 20; for 0.5 h;
Stage #2: with 4-methyl-morpholine;ammonium hydroxide in tetrahydrofuran;water at 20; for 18 h;
Steps:
57
To a solution of 2-amino-6-methoxybenzoic acid (9.0 g, 53.8 mmol) in THF (270 mL) was added 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide hydrochloride (23.7 g, 123.7 mmol) and hydroxybenzotriazole (23.9 g, 156.0 mmol). The mixture was stirred at rt for 30 min, then N-methylmorpholine (18.2 g, 180.3 mmol) and NH4OH ( 50 vol % in H2O, 36 mL) were added. The reaction mixture was then stirred for 18 h at rt. After that time the reaction was concentrated under reduced pressure and diluted with ethyl acetate (600 mL). The organic phase was washed with brine (2×100 mL), dried over Na2SO4 and concentrated under reduced pressure to give 2-amino-6-methoxybenzamide (8.80 g, 98%) as a light yellow solid: 1H NMR (400 MHz, DMSO-d6): δ 7.55 (br s, 1H), 7.29 (br s, 1H), 7.01 (t, J=8.4 Hz, 1H), 6.36 (br s, 2H), 6.30 (dd, J=8.4, 0.8 Hz, 1H), 6.17 (dd, J=8.0, 1.2 Hz, 1H), 3.76 (s, 3H)
References:
US2014/140956,2014,A1 Location in patent:Paragraph 0860
1591-37-3
73 suppliers
inquiry

1591-38-4
18 suppliers
inquiry

38469-85-1
20 suppliers
inquiry

1591-38-4
18 suppliers
inquiry
![5-methoxy-1H-benzo[d][1,3]oxazine-2,4-dione](/CAS/GIF/67765-42-8.gif)
67765-42-8
55 suppliers
$45.00/10mg

1591-38-4
18 suppliers
inquiry
![Carbamic acid, [2-(aminocarbonyl)-3-methoxyphenyl]-, 1,1-dimethylethyl ester (9CI)](/CAS/20210305/GIF/136247-99-9.gif)
136247-99-9
0 suppliers
inquiry

1591-38-4
18 suppliers
inquiry