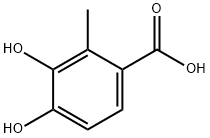
Benzoic acid, 3,4-dihydroxy-2-Methyl- synthesis
- Product Name:Benzoic acid, 3,4-dihydroxy-2-Methyl-
- CAS Number:168899-47-6
- Molecular formula:C8H8O4
- Molecular Weight:168.15
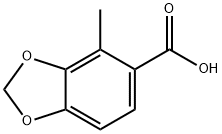
162506-58-3
19 suppliers
inquiry

168899-47-6
25 suppliers
inquiry
Yield: 82%
Reaction Conditions:
Stage #1:4-methyl-benzo[1,3]dioxole-5-carboxylic acid with boron trichloride in hexane;dichloromethane at 20; for 3 h;
Stage #2: with water in hexane;dichloromethane for 0.5 h;
Steps:
1.7
To a round bottom flask equipped with magnetic stirring, a 25% NaOH gas trap, and a nitrogen inlet was added 2-methylpiperonylic acid (26.0 g, 144 mmol) and methylene chloride (482 mL). To this was added the boron trichloride solution. The mixture was stirred at room temperature for 3 hours. The reaction was quenched by the careful addition of 500 mL of water. CAUTION: this step causes foaming and generates large amounts of HCl. The reaction was stirred for 30 min. The layers were separated, the TLC of the organic layer was checked to confirm the absence of product and the organic layer was subsequently discarded. The aqueous layer was extracted 3 times with ethyl acetate. The combined extracts were washed once with water, once with saturated NaCl solution, dried over sodium sulfate, filtered, and evaporated to give a tan solid, 3,4-dihydroxy-2-methylbenzoic acid, (20.0 g, 119 mmol) in 82% yield. Note: BCl3 in heptane has been used as well with equal success. 1H-NMR (300 MHz, CD3COCD3) δ (ppm): 2.51 (s, 3H), 6.76 (d, 1H), 7.45 (overlapping s+d, 1H+1H), 9.06 (s, 1H). TLC Rf=0.20 (1:1 ethyl acetate/hexane).
References:
Hormann, Robert Eugene;Tice, Colin M.;Chortyk, Orestes;Smith, Howard;Meteyer, Thomas US2005/209283, 2005, A1 Location in patent:Page/Page column 33
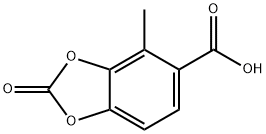
861559-98-0
0 suppliers
inquiry

168899-47-6
25 suppliers
inquiry

861559-98-0
0 suppliers
inquiry
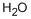
7732-18-5
462 suppliers
$12.69/100ml

168899-47-6
25 suppliers
inquiry