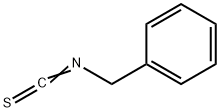
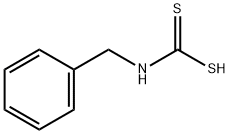
BENZYL ISOTHIOCYANATE synthesis
- Product Name:BENZYL ISOTHIOCYANATE
- CAS Number:622-78-6
- Molecular formula:C8H7NS
- Molecular Weight:149.21
75-15-0
260 suppliers
$40.00/100 g
100-46-9
495 suppliers
$5.00/5 g

622-78-6
253 suppliers
$12.00/1g
Yield:622-78-6 97%
Reaction Conditions:
Stage #1:carbon disulfide;benzylamine in dimethyl sulfoxide for 0.25 h;Inert atmosphere;
Stage #2: with N-benzyl-trimethylammonium hydroxide in dimethyl sulfoxide for 2 h;Inert atmosphere;
Steps:
General procedure
General procedure: To a solution of amines (1 mmol) in DMSO, added carbon disulphide under nitrogen atmosphere and stirred for 15 min to afford compound 2. Further added triton-B after reaction mixture was stirred for 2 h to afford compound 3 (Scheme-I). After completion, extracted with ethyl acetate to yield isothiocyanates up to 80-99 %. The synthesized trithiocarbonates was subjected to 1H NMR, 13C NMR and mass spectroscopy for structure elucidation. The novel one pot synthesis yields good to excellent amount of isothiocyanates, both cyclic and acyclic substituent at room temperature.
References:
Singh, Neha;Khare, Richa [Asian Journal of Chemistry,2019,vol. 31,# 7,p. 1636 - 1638]
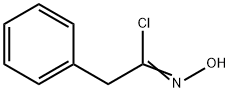
701-72-4
7 suppliers
inquiry

622-78-6
253 suppliers
$12.00/1g
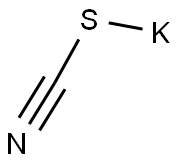
333-20-0
456 suppliers
$13.50/100G
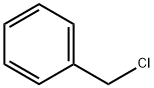
100-44-7
661 suppliers
$13.50/250G
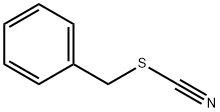
3012-37-1
128 suppliers
$14.00/1g

622-78-6
253 suppliers
$12.00/1g
15867-06-8
0 suppliers
inquiry

622-78-6
253 suppliers
$12.00/1g