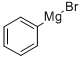beta-methylenephenethyl alcohol synthesis
- Product Name:beta-methylenephenethyl alcohol
- CAS Number:6006-81-1
- Molecular formula:C9H10O
- Molecular Weight:134.18
Yield:6006-81-1 80%
Reaction Conditions:
with copper(l) iodide in tetrahydrofuran;toluene at -78 - 20;Inert atmosphere;
Steps:
2-Phenylallyl alcohol
A suspension of CuI (494 mg, 2.60 mmol, 0.5 equiv) in toluene (6 mL) was cooled to -78 °C. Then, propargyl alcohol (300 μL, 291 mg, 5.19 mmol, 1.0 equiv) and PhMgBr (15.6 mL, 15.6 mmol, 1.0 Min THF, 3.0 equiv) were added, and the mixture was warmed to room temperature overnight. After the addition of NH4Cl solution (2 mL), the layers were separated, and the aqueous layer was extracted with EtOAc (3 × 10 mL). The combined organic layers were dried (Na2SO4), filtered, and the volatile components were removed in vacuo. The crude product was purified by flash chromatography (3 × 20 cm, Pn-EtOAc, 6:1), giving the desired alcohol as a light yellow oil (557 mg, 4.15 mmol, 80%); Rf= 0.38 (Pn-EtOAc, 4:1) [UV, KMnO4]. 1H NMR (500 MHz, CDCl3): δ = 4.56 (br s, 2 H, H-1), 5.36 (virt. q, 2J ≈4J = 1.3 Hz, 1 H, H-3), 5.47 (virt. q, 2J ≈4J = 1.1 Hz, 1 H, H-3), 7.30 (tt, 3J = 7.4 Hz, 4J = 1.5 Hz, 1 H, H-Arpara), 7.36 (virt. t, 3J ≈3J = 7.8 Hz, 2 H, H-Armeta), 7.45 (dd, 3J = 8.5 Hz, 4J = 1.5 Hz, 2 H, H-Arortho). 13C NMR (91 MHz, CDCl3): δ = 65.2 (t, C-1), 112.8 (t, C-3), 126.2 (d, C-Arortho), 128.1 (d, C-Arpara), 128.7 (d, C-Armeta), 138.6 (s, C-Aripso), 147.5 (s, C-2).
References:
Wegmann, Marcus;Bach, Thorsten [Synthesis,2017,vol. 49,# 1,art. no. SS-2016-Z0504-OP,p. 209 - 217] Location in patent:supporting information