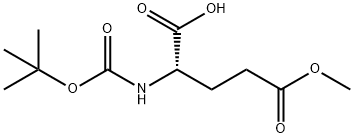
BOC-GLU(OME)-OH synthesis
- Product Name:BOC-GLU(OME)-OH
- CAS Number:45214-91-3
- Molecular formula:C11H19NO6
- Molecular Weight:261.27
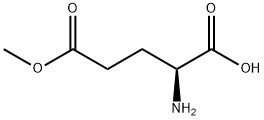
1499-55-4
274 suppliers
$6.00/5g

24424-99-5
821 suppliers
$13.50/25G

45214-91-3
183 suppliers
$10.00/5g
Yield:45214-91-3 100%
Reaction Conditions:
with triethylamine in 1,4-dioxane;water at 20; for 18 h;
Steps:
4.1 Step 1
In a round bottom flask, triethylamine (188g, 1.86mol, l .Oeq) was added dropwise to a stirred solution of di-tert-butyl dicarbonate (162g, 0.744mol, 1.2eq) and compound A4-1 (lOOg, 0.62mol, l .Oeq) in water (500mL) and 1,4-dioxane (500mL). After stirring for 18hrs at room temperature, the solution was extracted with MTBE (500mL*2) and the aqueous phase was cooled on ice and carefully acidified to pH 3 by slow addition of 10% citric acid solution. The urethane was then extracted twice with ethyl acetate, and the combined extracts was washed with brine, dried over anhydrous sodium sulfate, and concentrated to give compound A4-2 as clear viscous oil (180g, yield 100%). MS-ESI:[M+1]+: 262.1
References:
LIANG, Congxin WO2018/67422, 2018, A1 Location in patent:Page/Page column 47
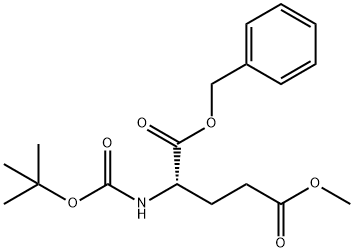
132245-78-4
14 suppliers
inquiry

45214-91-3
183 suppliers
$10.00/5g

24424-99-5
821 suppliers
$13.50/25G
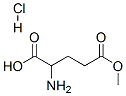
3077-51-8
8 suppliers
inquiry

45214-91-3
183 suppliers
$10.00/5g

1499-55-4
274 suppliers
$6.00/5g

45214-91-3
183 suppliers
$10.00/5g