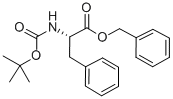
BOC-PHE-OBZL synthesis
- Product Name:BOC-PHE-OBZL
- CAS Number:66617-58-1
- Molecular formula:C21H25NO4
- Molecular Weight:355.43
Yield:66617-58-1 99%
Reaction Conditions:
with potassium carbonate in acetone;Heating / reflux;
Steps:
1.19(i)
Synthesis of Compound 19; To Boc-(L)-Phe-OH (265 mg, 1.00 mmol) and potassium carbonate (691 mg, 5.0 mmol) in acetone (30 ml) was added benzyl bromide (0.24 ml, 2.0 mmol). The resulting solution was heated at reflux overnight before being cooled, filtered and evaporated to dryness. The resultant residue was subjected to flash column chromatography over silica, eluting with 5% ethyl acetate/hexane to first remove benzyl bromide, then with DCM to yield the product 19(i) as a white solid (351 mg, 99%).1H NMR (500 MHz, CDCl3) δ 1.46, s, 9H; 3.12, m, 2H; 4.68, m, IH; 5.15, ABq, J = 12.3 Hz, IH; 5.16, m, NH; 5.19, ABq, / = 12.3 Hz, IH; 7.09, m, 2H; 7.25, m, 3H; 7.32, m, 2H; 7.36, m, 2H. MS (ES +ve) m/z 401.1 (40%) [M+HCOOH]+; 378.1 (20) [M+Na]+; 356.1 (25) [M+H]+; 300.0 (60) [M+H-C+Hsf; 256.0 (100) [M+H-BOC]"1".
References:
WO2006/74501,2006,A1 Location in patent:Page/Page column 79

13734-34-4
536 suppliers
$5.50/5g
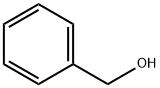
100-51-6
1379 suppliers
$5.00/100g

66617-58-1
61 suppliers
inquiry

13734-34-4
536 suppliers
$5.50/5g
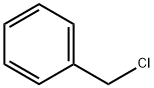
100-44-7
631 suppliers
$13.50/250G

66617-58-1
61 suppliers
inquiry
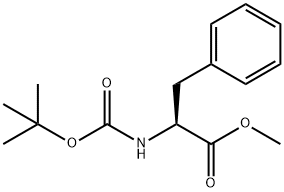
51987-73-6
211 suppliers
$9.00/5g

100-51-6
1379 suppliers
$5.00/100g

66617-58-1
61 suppliers
inquiry
![1-Propanone, 3-(diphenylphosphinyl)-1-[3-(phenylmethyl)-2-triazen-1-yl]-](/CAS/20210305/GIF/1155946-51-2.gif)